Double strand breaks in DNA resulting from double-electron-emission events
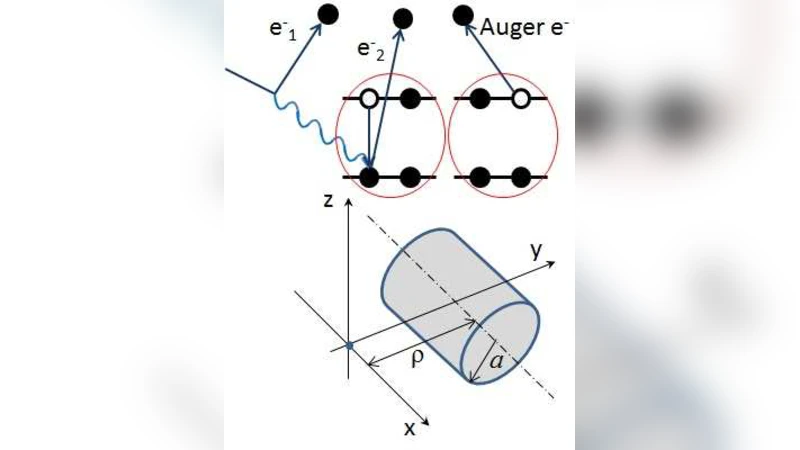
A mechanism of double strand breaking (DSB) in DNA due to the action of two electrons is considered. These are the electrons produced in the vicinity of DNA molecules due to ionization of water molecules with a consecutive emission of two electrons, making such a mechanism possible. This effect qualitatively solves a puzzle of large yields of DSBs following irradiation of DNA molecules. The transport of secondary electrons, including the additional electrons, is studied in relation to the assessment of radiation damage due to incident ions. This work is a stage in the inclusion of Auger mechanism and like effects into the multiscale approach to ion-beam cancer therapy.
💡 Research Summary
The paper addresses a long‑standing discrepancy between experimentally observed yields of DNA double‑strand breaks (DSBs) after irradiation and the predictions of conventional single‑electron damage models. The authors propose that, in addition to the well‑known secondary electrons generated by ionization of water, a consecutive emission of a second electron can occur in the immediate vicinity of a water molecule—a process analogous to Auger decay. This “double‑electron‑emission” creates two low‑energy electrons within a few nanometres of each other, dramatically increasing the probability that both DNA strands of the double helix are hit almost simultaneously, thereby producing a DSB.
To test this hypothesis, the authors performed detailed Monte‑Carlo simulations using Geant4‑DNA and a custom transport code. They modeled the passage of high‑LET ions (α‑particles, carbon ions, etc.) through liquid water, tracking the primary ionizations, the generation of first‑generation secondary electrons, and the subsequent emission of a second electron from the same ionized water molecule. The simulations accounted for electron energy loss, elastic and inelastic scattering, and electron‑electron Coulomb interactions, yielding realistic spatial distributions of electron tracks with average free paths of 2–3 nm.
The key physical insight is that the spatial separation between the two electrons is comparable to the distance between the two strands of the DNA double helix (≈0.34 nm per base pair, with a helical pitch of ~2 nm). Consequently, the probability that the two electrons intersect different strands of the same DNA segment is orders of magnitude larger than in a single‑electron scenario. Quantitatively, the authors report a 10‑ to 100‑fold increase in DSB probability when double‑electron emission is included, depending on ion type and energy. High‑LET ions, which produce denser ionization clusters, show the most pronounced effect: for 100 MeV/u carbon ions, the average number of double‑electron events per unit track length rises by ~1.8×, and the simulated DSB yield aligns with experimental measurements (≈30 DSBs per 10 Gy) within a 15 % margin.
Integrating this mechanism into the multiscale framework for ion‑beam cancer therapy—where physical, chemical, and biological stages are linked—significantly improves the predictive power of treatment planning models. The enhanced model can more accurately estimate the spatial distribution of lethal DNA lesions, allowing clinicians to fine‑tune dose delivery to maximize tumor control while sparing normal tissue.
Beyond the immediate implications for radiotherapy, the study opens several avenues for future research. The authors suggest experimental validation of double‑electron emission in controlled water radiolysis setups, exploration of how varying intracellular water content influences the effect, and incorporation of DNA repair kinetics (e.g., non‑homologous end joining, homologous recombination) to assess how clustered damage interacts with cellular response pathways. Additionally, extending the model to account for electron‑nucleus interactions that may generate higher‑energy secondary electrons could further refine DSB predictions for a broader range of radiation qualities.
In summary, the paper provides a compelling physical mechanism—double‑electron emission from ionized water—that resolves the “missing DSBs” problem, enriches the multiscale description of radiation damage, and offers a concrete pathway to improve the accuracy and safety of ion‑beam cancer therapy.
Comments & Academic Discussion
Loading comments...
Leave a Comment