Phenomenological analysis of ATP dependence of motor protein
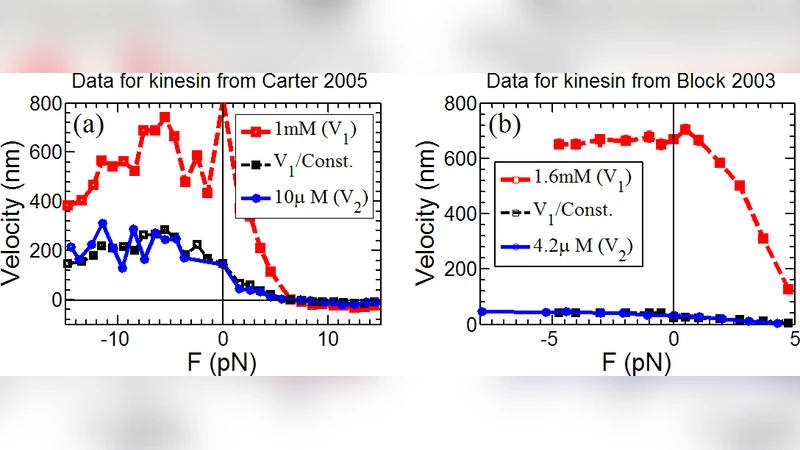
In this study, through phenomenological comparison of the velocity-force data of processive motor proteins, including conventional kinesin, cytoplasmic dynein and myosin V, we found that, the ratio between motor velocities of two different ATP concentrations is almost invariant for any substall, superstall or negative external loads. Therefore, the velocity of motor can be well approximated by a Michaelis-Menten like formula $V=\atp k(F)L/(\atp +K_M)$, with $L$ the step size, and $k(F)$ the external load $F$ dependent rate of one mechanochemical cycle of motor motion in saturated ATP solution. The difference of Michaelis-Menten constant $K_M$ for substall, superstall and negative external load indicates, the ATP molecule affinity of motor head for these three cases are different, though the expression of $k(F)$ as a function of $F$ might be unchanged for any external load $F$. Verifications of this Michaelis-Menten like formula has also been done by fitting to the recent experimental data.
💡 Research Summary
In this paper the authors present a phenomenological analysis of how ATP concentration and external mechanical load jointly determine the velocity of three well‑studied processive motor proteins: conventional kinesin, cytoplasmic dynein, and myosin V. By gathering published velocity–force (V–F) data measured at a range of ATP concentrations—from millimolar (saturating) down to micromolar levels—the authors first observe a striking regularity: for any given load (whether sub‑stall, super‑stall, or even assisting/negative), the low‑ATP velocity curve can be obtained by scaling the high‑ATP curve by a nearly constant factor. In the original figures this factor is typically between 2 and 30 depending on the motor, the load regime, and the specific dataset, but it remains essentially independent of the exact force value within each regime.
Motivated by this observation, the authors propose a simple Michaelis–Menten‑type expression for the motor velocity:
\
Comments & Academic Discussion
Loading comments...
Leave a Comment