Mechanisms of kinetic trapping in self-assembly and phase transformation
In self-assembly processes, kinetic trapping effects often hinder the formation of thermodynamically stable ordered states. In a model of viral capsid assembly and in the phase transformation of a lattice gas, we show how simulations in a self-assembling steady state can be used to identify two distinct mechanisms of kinetic trapping. We argue that one of these mechanisms can be adequately captured by kinetic rate equations, while the other involves a breakdown of theories that rely on cluster size as a reaction coordinate. We discuss how these observations might be useful in designing and optimising self-assembly reactions.
💡 Research Summary
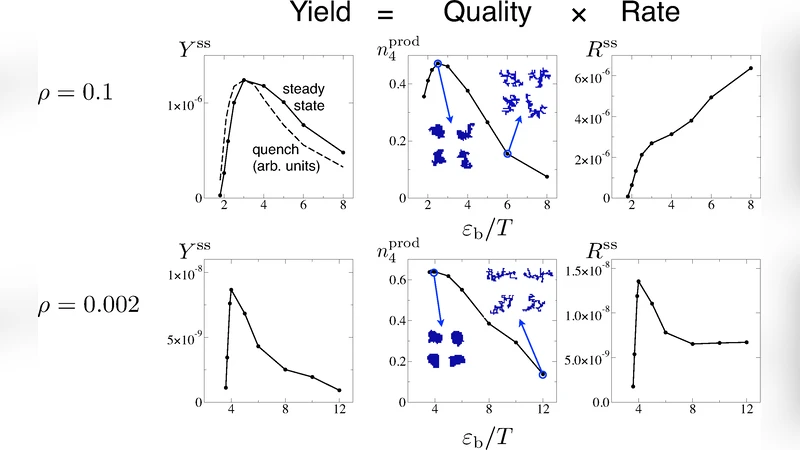
The paper investigates kinetic trapping in self‑assembly by studying two representative systems: an empty icosahedral viral capsid model and a two‑dimensional lattice‑gas model that undergoes phase separation. Both systems are simulated under conventional NVT conditions and under a novel steady‑state ensemble in which large clusters are periodically removed, catalogued as “products,” and the removed particles are re‑introduced as free monomers. This steady‑state protocol yields time‑independent averages of two key observables: the production rate R_ss (clusters formed per unit time and volume) and the product quality Q_prod (fraction of products that are perfectly assembled – complete capsids in the viral model, or particles with four bonds in the lattice gas). The overall yield Y_ss = R_ss × Q_prod therefore captures the combined effect of kinetic speed and structural fidelity.
In the capsid model, subunits are rigid bodies with anisotropic interactions characterized by a dimensionless binding energy ε_b/T. NVT simulations show a classic non‑monotonic yield: weak binding gives insufficient thermodynamic drive, while overly strong binding leads to rapid formation of disordered aggregates that lack the precise geometry of a capsid. The steady‑state results reproduce this trend: as ε_b/T increases, R_ss rises monotonically, but Q_prod falls sharply once ε_b/T exceeds ≈5, producing a peak in Y_ss at intermediate binding strength. The kinetic trap here is not a depletion of free subunits but the emergence of large, malformed clusters that dominate the product stream.
The lattice‑gas model, defined by nearest‑neighbour bonds of energy ε_b, exhibits analogous behavior. At a relatively high density (ρ = 0.1), increasing ε_b/T boosts R_ss while degrading Q_prod, again yielding a non‑monotonic Y_ss. At a low density (ρ = 0.002), however, R_ss itself becomes non‑monotonic: strong bonds cause rapid cluster nucleation, which quickly consumes free particles and leads to a “stalling” regime where further growth is limited by monomer scarcity. This illustrates a second kinetic‑trapping mechanism, previously described by Zlotnick for viral capsids, in which the production rate collapses because the system is starved of building blocks.
To differentiate these mechanisms, the authors introduce a “cluster equilibration” condition. Clusters are indexed by size n and morphology α; equilibrium requires the relative populations of different morphologies at fixed n to follow a Boltzmann distribution, i.e., N_{n,α}/N_{n,γ}=exp
Comments & Academic Discussion
Loading comments...
Leave a Comment