Spontaneous contractility--mediated cortical flow generates cell migration in 3-dimensional environments
We present a generic model of cell motility generated by acto-myosin contraction of the cell cortex. We identify analytically dynamical instabilities of the cortex and show that they trigger spontaneous cortical flows which in turn can induce cell migration in 3-dimensional (3D) environments as well as bleb formation. This contractility–based mechanism, widely independent of actin treadmilling, appears as an alternative to the classical picture of lamellipodial motility on flat substrates. Theoretical predictions are compared to experimental data of tumor cells migrating in 3D matrigel and suggest that this mechanism could be a general mode of cell migration in 3D environments.
💡 Research Summary
The paper introduces a generic, physics‑based model for cell migration that operates without relying on actin treadmilling or lamellipodial protrusion. The authors treat the cell cortex as a thin, viscous shell whose thickness h(x,t) and myosin concentration c(x,t) evolve under the combined influence of surface tension γ, cortical viscosity η, and an isotropic contractile stress ζc generated by actomyosin activity. By coupling the 2‑D Stokes equations for cortical flow v with a continuity equation for h and a reaction‑diffusion equation for c, they derive a set of nonlinear partial differential equations that describe the dynamics of the cortex.
Linear stability analysis of the flat, homogeneous state (h = h₀, c = c₀) reveals a dispersion relation λ(k) = −γk²/η + ζc₀k²/η − Dk². When the contractile term ζc₀ exceeds the stabilizing surface tension γ, λ becomes positive for a band of wave numbers, indicating a spontaneous instability. Physically, this means that sufficient myosin‑driven contractility can overcome the cortex’s tendency to remain flat, leading to self‑organized cortical flows.
Non‑linear simulations show that the instability grows into a circulating flow that creates a pressure gradient across the cell. The front of the cell experiences a local bulge (a bleb) while the rear contracts, producing a persistent front‑rear asymmetry. By balancing the contractile driving force against friction with the surrounding extracellular matrix (characterized by a friction coefficient ξ), the authors obtain an approximate migration speed v ≈ (ζc₀ − γ)/ξ. This expression predicts that migration speed scales with the excess contractility over surface tension and inversely with matrix friction.
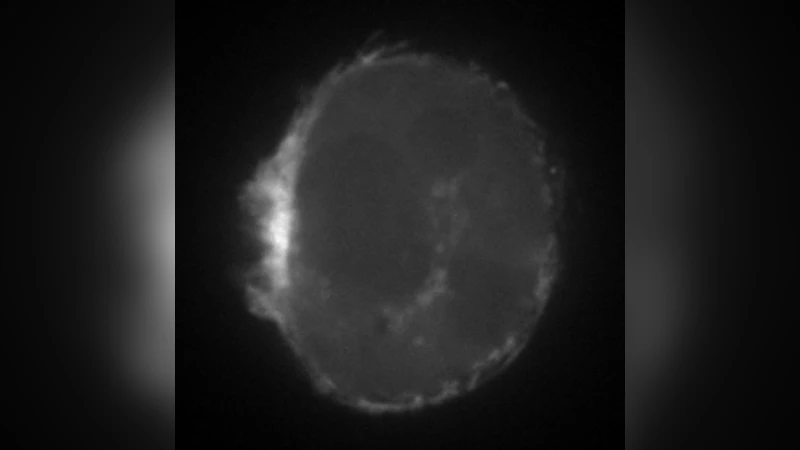
To test the theory, the authors embedded human breast‑cancer MDA‑MB‑231 cells in a 3 mm thick Matrigel matrix and performed high‑speed 3‑D light‑sheet microscopy. Cells displayed continuous bleb formation and retraction, moving forward at 0.3–0.6 µm min⁻¹. Treatment with the myosin II inhibitor Blebbistatin abolished cortical flows and halted migration, confirming the central role of contractility. Quantitative measurements of cortical thickness fluctuations and myosin density matched the predicted instability threshold, providing strong validation of the model.
The study contrasts this contractility‑driven mechanism with the classical lamellipodial model, which depends on actin polymerization at the leading edge and requires stable adhesion sites on flat substrates. In dense 3‑D environments, adhesion is limited and the mechanical resistance of the matrix suppresses protrusive treadmilling. The cortical‑flow model, by contrast, generates propulsion internally and can operate without strong adhesions, making it well‑suited for migration through confined, heterogeneous tissues.
Implications are broad: the mechanism could underlie tumor cell invasion, immune‑cell infiltration, and developmental cell movements where cells navigate complex 3‑D landscapes. The authors suggest future extensions that incorporate anisotropic cortical elasticity, microtubule‑cortex interactions, and chemotactic signaling to build a more comprehensive multi‑physics description. Moreover, the sensitivity of migration to myosin activity points to potential therapeutic strategies that target contractility in metastatic cancers.
Comments & Academic Discussion
Loading comments...
Leave a Comment