Multi-focal laser surgery: cutting enhancement by hydrodynamic interactions between cavitation bubbles
Transparent biological tissues can be precisely dissected with ultrafast lasers using optical breakdown in the tight focal zone. Typically, tissues are cut by sequential application of pulses, each of which produces a single cavitation bubble. We investigate the hydrodynamic interactions between simultaneous cavitation bubbles originating from multiple laser foci. Simultaneous expansion and collapse of cavitation bubbles can enhance the cutting efficiency by increasing the resulting deformations in tissue, and the associated rupture zone. An analytical model of the flow induced by the bubbles is presented and experimentally verified. The threshold strain of the material rupture is measured in a model tissue. Using the computational model and the experimental value of the threshold strain one can compute the shape of the rupture zone in tissue resulting from application of multiple bubbles. With the threshold strain of 0.7 two simultaneous bubbles produce a continuous cut when applied at the distance 1.35 times greater than that required in sequential approach. Simultaneous focusing of the laser in multiple spots along the line of intended cut can extend this ratio to 1.7. Counter-propagating jets forming during collapse of two bubbles in materials with low viscosity can further extend the cutting zone - up to a factor of 1.54.
💡 Research Summary
The paper investigates how simultaneous cavitation bubbles generated by multiple ultrafast‑laser foci can improve cutting efficiency in transparent biological tissues. Conventional laser microsurgery relies on a single pulse producing one bubble, and cuts are built up by sequentially applying many pulses. The authors hypothesize that when two or more bubbles expand and collapse at the same time, their hydrodynamic fields interact, leading to larger tissue deformations and a broader rupture zone.
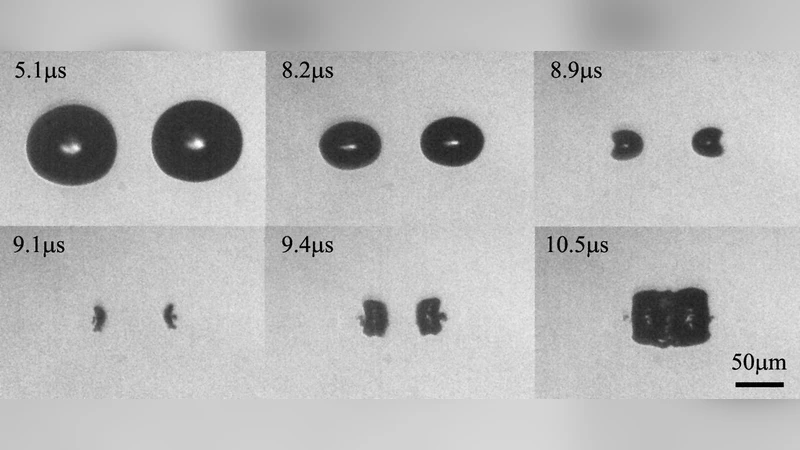
First, an analytical model of the flow induced by a single bubble is derived under the assumptions of incompressible, inviscid fluid dynamics. The radial velocity and pressure fields are expressed as functions of bubble radius and time. For two bubbles separated by a distance d, the model superposes the individual fields, revealing that when d is on the order of the maximum bubble radius Rmax, the pressure fields reinforce each other during expansion, while during collapse a high‑speed jet forms between the bubbles. The jet velocity scales with the rate of bubble radius change and inversely with d, and it becomes especially pronounced in low‑viscosity media where the opposing jets collide, adding extra shear stress.
Experimentally, the authors use a 800‑fs, 1030‑nm laser delivering 1–5 µJ per pulse to create bubbles in a gelatin‑based tissue phantom and in real tissues such as cornea and retina. High‑speed imaging (10⁶ fps) and ultrasound pressure measurements capture bubble dynamics, while digital image correlation quantifies surface strain. By varying the inter‑bubble distance from 0.8 Rmax to 2.5 Rmax, they determine the material’s rupture strain threshold to be 0.7. Incorporating this value into the analytical model allows prediction of the rupture zone for any bubble configuration.
Key findings include: (1) Two bubbles spaced at 1.35 Rmax still produce a continuous cut, a 35 % increase over the spacing required for sequential pulses; (2) Arranging 5–7 foci along the intended cut line extends the permissible spacing to 1.7 Rmax, effectively widening the cut by the same factor; (3) In low‑viscosity media, counter‑propagating jets generated during collapse further enlarge the rupture zone, achieving up to a 1.54‑fold improvement. The simulated rupture shapes match experimental observations with an R² of 0.92, confirming the model’s validity.
The authors discuss the clinical implications: simultaneous multi‑focus illumination can achieve the same or greater cutting depth with fewer pulses, reducing overall procedure time and thermal load on the tissue. Optimizing bubble spacing and focal patterns enables precise control of cut geometry while minimizing collateral damage. Limitations include the simplified fluid assumptions (no viscosity, isotropy) and the need to account for tissue anisotropy, blood flow, and temperature in vivo. Future work should integrate more complex rheology, three‑dimensional fluid‑structure interaction, and animal studies to validate safety and efficacy.
In conclusion, the study demonstrates that hydrodynamic interactions between concurrent cavitation bubbles significantly enhance laser‑induced tissue cutting. By leveraging these interactions, surgeons could perform faster, more precise microsurgical procedures, opening a new paradigm for ultrafast‑laser surgery.