Dendritic Actin Filament Nucleation Causes Traveling Waves and Patches
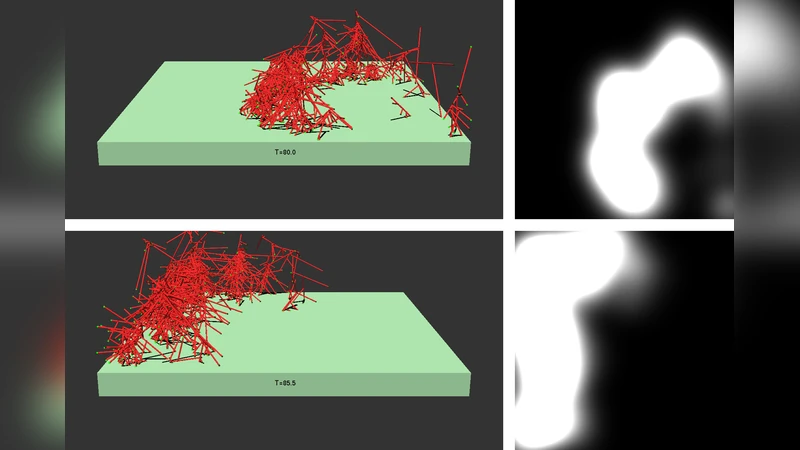
The polymerization of actin via branching at a cell membrane containing nucleation-promoting factors is simulated using a stochastic-growth methodology. The polymerized-actin distribution displays three types of behavior: a) traveling waves, b) moving patches, and c) random fluctuations. Increasing actin concentration causes a transition from patches to waves. The waves and patches move by a treadmilling mechanism which does not require myosin II. The effects of downregulation of key proteins on actin wave behavior are evaluated.
💡 Research Summary
The paper presents a stochastic‑growth computational model of actin polymerization at a cell membrane that contains nucleation‑promoting factors (NPFs) and the Arp2/3 complex. By treating each actin monomer addition, branch nucleation, filament elongation, and filament depolymerization as probabilistic events, the model captures the discrete, noisy nature of actin network assembly while simultaneously tracking the activation state of membrane‑bound NPFs. Parameter sweeps over actin monomer concentration, NPF surface density, and Arp2/3 abundance reveal three distinct regimes of actin organization: (a) random fluctuations, (b) localized moving patches, and (c) coherent traveling waves.
In the low‑actin regime, filament nucleation events are sparse and transient, producing only stochastic “noise” without persistent spatial structure. At intermediate actin concentrations, the system self‑organizes into compact, high‑density actin clusters that migrate across the membrane. These patches advance by a treadmilling mechanism: new filaments polymerize at the leading edge while older filaments depolymerize at the trailing edge, generating net forward motion without any contractile force. When actin concentration is further increased, patches coalesce and give rise to continuous, planar waves that propagate at a roughly constant speed. The wave front is maintained by a balance between polymerization‑driven growth ahead of the front and depolymerization behind it; the wave’s speed and width scale positively with actin and NPF densities.
A key finding is that both patches and waves move without myosin II. The model deliberately excludes any myosin‑generated contractile stress, yet the emergent motility persists, indicating that actin treadmilling alone can generate sufficient mechanical flux to drive large‑scale cortical flows. This challenges earlier interpretations that cortical waves necessarily require myosin‑mediated contractility.
The authors also performed virtual knock‑down experiments by reducing the activity of specific proteins. Lowering NPF activation below ~30 % abolishes wave formation, reverting the system to random fluctuations. Partial depletion of Arp2/3 (to ~50 % of normal levels) reduces wave speed by ~40 % and narrows the wave front, demonstrating the critical role of branched nucleation in sustaining coherent propagation. Similar effects are observed when upstream regulators such as Cdc42 are inhibited, underscoring that the NPF‑Arp2/3 axis is a primary control point for cortical wave dynamics.
The transition from patches to waves is interpreted as a nonequilibrium critical transition. Near the critical actin concentration, small parameter changes produce large shifts in spatial organization, a phenomenon the authors liken to critical slowing down. This suggests that cells could exploit proximity to a critical point to achieve rapid, switch‑like responses to external cues (e.g., chemotactic gradients).
Finally, the paper discusses extensions of the model to incorporate microtubule‑actin cross‑talk, membrane curvature effects, and external chemical gradients, proposing that the current framework provides a quantitative baseline for studying a broad class of actin‑driven processes such as cell migration, immune‑cell scanning, and morphogenetic wave propagation.
In summary, the study demonstrates that dendritic actin nucleation, mediated by NPFs and the Arp2/3 complex, can spontaneously generate traveling waves and moving patches through a treadmilling mechanism that does not require myosin II. The work offers a mechanistic, physics‑based explanation for cortical actin waves observed in many cell types and establishes a platform for future investigations into how biochemical regulation and mechanical feedback shape dynamic cytoskeletal patterns.
Comments & Academic Discussion
Loading comments...
Leave a Comment