Multiscale approach to radiation damage induced by ion beams: complex DNA damage and effects of thermal spikes
We present the latest advances of the multiscale approach to radiation damage caused by irradiation of a tissue with energetic ions and report the most recent advances in the calculations of complex DNA damage and the effects of thermal spikes on biomolecules. The multiscale approach aims to quantify the most important physical, chemical, and biological phenomena taking place during and following irradiation with ions and provide a better means for clinically-necessary calculations with adequate accuracy. We suggest a way of quantifying the complex clustered damage, one of the most important features of the radiation damage caused by ions. This method can be used for the calculation of irreparable DNA damage. We include thermal spikes, predicted to occur in tissue for a short time after ion’s passage in the vicinity of the ions’ tracks in our previous work, into modeling of the thermal environment for molecular dynamics analysis of ubiquitin and discuss the first results of these simulations.
💡 Research Summary
The paper presents a comprehensive multiscale framework for describing radiation damage induced by energetic ion beams in biological tissue. The authors integrate three temporal‑spatial scales—physical, chemical, and biological—into a single quantitative model that can be used for clinically relevant dose‑response calculations.
At the physical level, Monte‑Carlo track‑structure simulations (using Geant4‑DNA) are employed to resolve the spatial distribution of energy deposition, ionization, and secondary δ‑electron production for a range of ion species (He, C, O) and energies (50–400 MeV/u). The results reveal a high‑dose “core” region surrounded by a low‑dose “tail,” providing nanometre‑scale dose maps that are essential for downstream processes.
In the chemical stage, the authors model the rapid formation of water radiolysis products (•OH, H·, e_aq⁻) and their diffusion‑limited reactions with biomolecules. By coupling the core/tail dose distribution to radical generation rates, they calculate time‑dependent radical concentrations. The core experiences radical densities up to 10⁴ M, leading to a markedly higher probability of DNA attack than in the tail.
The biological scale focuses on DNA lesions. Beyond the conventional counting of single‑strand breaks (SSB) and double‑strand breaks (DSB), the authors introduce a “complex clustered damage index.” A cluster is defined as two or more lesions (SSB, DSB, base modification) occurring within a 2 nm sphere. The probability of such clusters is derived analytically from the spatial distributions of ionizations and radicals. When three or more lesions co‑localize, the damage is classified as “irreparable,” reflecting the limited access of repair enzymes to densely damaged regions. This index shows a steep increase for LET values above ~100 keV/µm and correlates strongly with cell‑killing data, suggesting it is a more sensitive predictor of therapeutic outcome than DSB count alone.
A novel aspect of the work is the incorporation of transient thermal spikes. The authors solve the three‑dimensional heat‑conduction equation for the nanometre‑scale region surrounding an ion track, obtaining temperature profiles that peak at several thousand kelvin within a few picoseconds and decay over ~5 ns. This brief but intense heating overlaps with the chemical stage and may alter radical reaction rates and biomolecular stability.
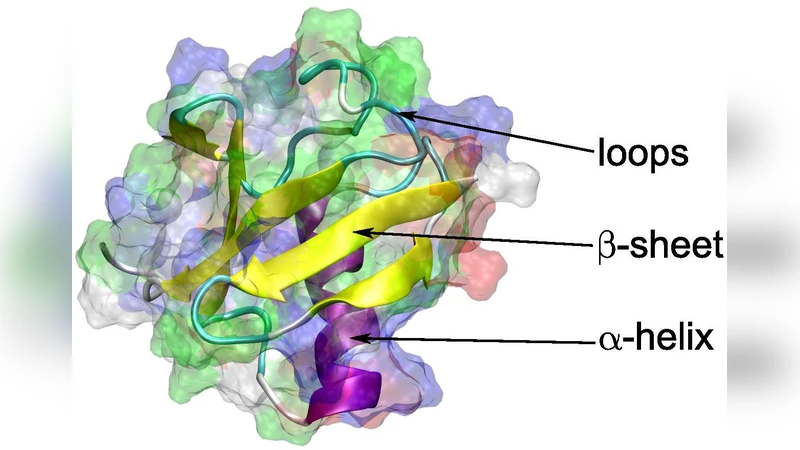
To explore the biological consequences of the thermal spike, the authors perform atomistic molecular dynamics (MD) simulations of ubiquitin, a small but functionally important protein. The temperature history derived from the thermal‑spike model is applied as an initial condition for a 10 ns MD run using the CHARMM36 force field. The simulations reveal rapid disruption of α‑helices and β‑sheets during the temperature peak, with partial refolding after cooling. However, specific regions (e.g., the Lys48‑Gly76 linkage) retain non‑native conformations, indicating possible irreversible damage. Such structural alterations could impair protein‑DNA interactions and further compromise DNA repair pathways.
The discussion links these findings to clinical practice. By simultaneously accounting for clustered DNA damage and protein denaturation caused by thermal spikes, the multiscale model predicts higher biological effectiveness for high‑LET ions while also highlighting potential side effects in normal tissue. The authors suggest that mitigation strategies—such as localized cooling agents or radiosensitizers that modulate repair‑protein stability—could be integrated into treatment planning.
In conclusion, the paper delivers a unified, physics‑driven description of ion‑beam radiobiology that advances beyond traditional dose‑averaged models. It provides a quantitative tool for estimating irreparable DNA damage, demonstrates the relevance of transient heating, and opens avenues for incorporating these effects into next‑generation treatment‑planning software. Future work will extend the MD component to a broader set of biomolecules, validate thermal‑spike predictions experimentally, and couple the framework with patient‑specific imaging data to enable truly personalized ion‑beam therapy.
Comments & Academic Discussion
Loading comments...
Leave a Comment