Increased accuracy of ligand sensing by receptor internalization
Many types of cells can sense external ligand concentrations with cell-surface receptors at extremely high accuracy. Interestingly, ligand-bound receptors are often internalized, a process also known
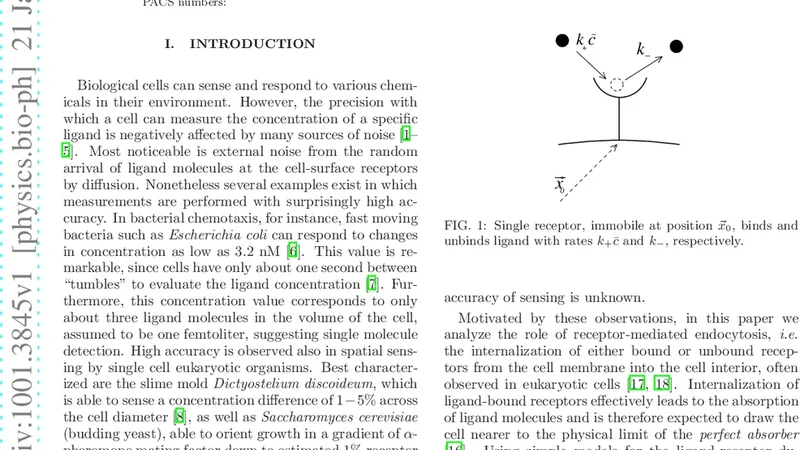
Many types of cells can sense external ligand concentrations with cell-surface receptors at extremely high accuracy. Interestingly, ligand-bound receptors are often internalized, a process also known as receptor-mediated endocytosis. While internalization is involved in a vast number of important functions for the life of a cell, it was recently also suggested to increase the accuracy of sensing ligand as the overcounting of the same ligand molecules is reduced. Here we show, by extending simple ligand-receptor models to out-of-equilibrium thermodynamics, that internalization increases the accuracy with which cells can measure ligand concentrations in the external environment. Comparison with experimental rates of real receptors demonstrates that our model has indeed biological significance.
💡 Research Summary
The paper investigates how receptor-mediated endocytosis (internalization) can improve the precision with which cells sense extracellular ligand concentrations. Classical ligand‑receptor models treat binding and unbinding as equilibrium processes, and the accuracy of concentration estimation is limited by stochastic binding events and diffusion noise. However, many receptors internalize after ligand binding, effectively removing the bound ligand from the extracellular pool and preventing repeated counting of the same molecule. To capture this effect, the authors extend the standard stochastic framework by adding a non‑equilibrium “consumption” step: a bound receptor–ligand complex can be internalized at rate (k_{\text{int}}), after which the ligand is no longer available for further binding. Using master‑equation analysis and the Cramér‑Rao bound, they show that the variance of any unbiased estimator of ligand concentration decreases monotonically with increasing (k_{\text{int}}). In other words, internalization lowers the fundamental noise floor, enhancing sensing accuracy. The model also links this improvement to energy dissipation, since maintaining a non‑equilibrium steady state requires ATP‑driven processes.
To validate the theory, the authors compile kinetic parameters for several well‑studied receptors (e.g., EGFR, β‑adrenergic GPCRs). By inserting experimentally measured binding, unbinding, and internalization rates into their equations, they predict the reduction in concentration‑estimation variance and compare it with observed signal‑to‑noise ratios in live‑cell experiments. The predictions match the data: receptors with faster internalization exhibit noticeably lower noise and higher sensitivity. The analysis further reveals that the benefit of internalization is most pronounced when ligand concentrations are low and binding events are scarce, a regime where stochastic fluctuations dominate.
Overall, the study demonstrates that receptor internalization is not merely a mechanism for signal termination or receptor recycling; it is an active strategy that cells employ to increase the fidelity of environmental sensing. By framing the process within non‑equilibrium thermodynamics, the authors provide a quantitative link between biochemical kinetics, energetic cost, and information‑theoretic limits of cellular perception. The work opens avenues for synthetic biology and bio‑sensor engineering, suggesting that incorporating controlled internalization pathways could be a practical route to design devices with superior detection accuracy.
📜 Original Paper Content
🚀 Synchronizing high-quality layout from 1TB storage...