Doping graphene by adsorption of polar molecules at the oxidized zigzag edges
We have theoretically investigated the electronic and magnetic properties of graphene whose zigzag edges are oxidized. The alteration of these properties by adsorption of $\mathrm{H_{2}O}$ and $\mathrm{NH_3}$ molecules have been considered. It was found that the adsorbed molecules form a cluster along the oxidized zigzag edges of graphene due to interaction with the electro-negative oxygen. Graphene tends to donate a charge to the adsorbates through the oxygen atoms and the efficiency of donation depends on the intermolecular distance and on the location of the adsorbed molecules relative to the plane of graphene. It was found that by appropriate selection of the adsorbates, a controllable and gradual growth of $p$-doping in graphene with a variety of adsorbed molecules can be achieved.
💡 Research Summary
The paper presents a comprehensive theoretical investigation of how the electronic and magnetic properties of graphene nanoribbons with oxidized zigzag edges (ox‑ZGNR) are altered by the adsorption of polar molecules, specifically water (H₂O) and ammonia (NH₃). Using spin‑polarized density‑functional theory (DFT) with the PBE‑GGA functional and D3 dispersion corrections, the authors model an eight‑zigzag‑unit‑wide nanoribbon whose edge carbon atoms are functionalized with carbonyl (C=O) and hydroxyl (C‑OH) groups, thereby creating an “oxidized” edge. The oxidation itself modifies the pristine edge states: the oxygen atoms draw electron density away from the edge carbons, suppressing the characteristic flat bands and inducing a modest band gap (~0.2 eV) together with a localized antiferromagnetic spin pattern on the edge atoms.
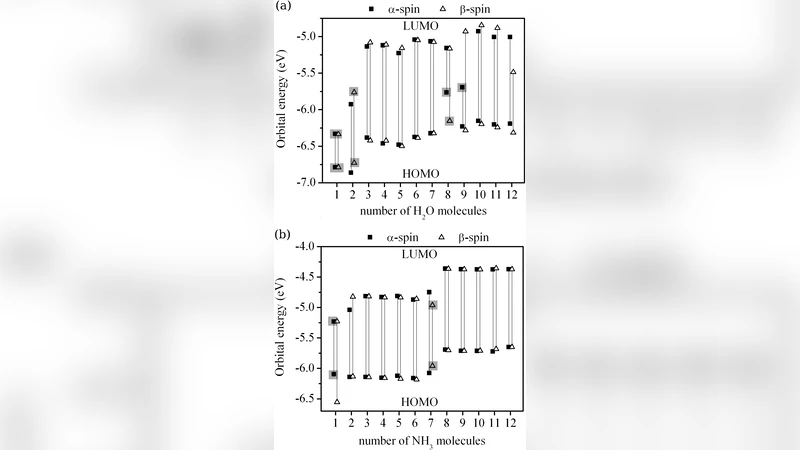
Adsorption of H₂O and NH₃ is then examined for 1–4 molecules placed along the oxidized edge. The molecules bind through hydrogen‑bond interactions with the edge oxygen atoms (H···O for water, N–H···O for ammonia) with binding energies of roughly 0.2 eV per molecule, leading to the formation of linear clusters that follow the edge contour. The authors systematically vary the intermolecular distance (d) and the tilt angle (θ) of the adsorbates relative to the graphene plane. When d ≤ 2.5 Å, charge transfer from the graphene to the adsorbates becomes pronounced. Bader charge analysis shows that a single water molecule extracts about 0.12 e from the ribbon, while a cluster of four water molecules can draw up to 0.35 e. Ammonia exhibits a comparable trend, especially when the molecule is oriented nearly perpendicular to the plane, maximizing overlap with the edge oxygen’s lone‑pair orbitals.
The transferred charge manifests as p‑type doping. The band structure evolves from a narrow gap semiconductor to a more pronounced p‑doped system: the Fermi level shifts toward the valence band, the gap widens to roughly 0.5 eV, and the density of states near the Fermi energy is dominated by hole‑like states. Spin density maps reveal that the local magnetic moments on the edge atoms are partially quenched by the adsorbates, yet the overall antiferromagnetic ordering persists. The authors attribute the doping mechanism primarily to the hydrogen‑bond network that mediates electron flow from the carbon‑oxygen framework to the polar molecules.
A key insight is that the efficiency of charge donation is not solely a function of the molecule’s intrinsic electronegativity; rather, it is strongly modulated by geometric factors—inter‑molecular spacing, adsorption height, and orientation. Consequently, by selecting different polar adsorbates and controlling their arrangement, one can achieve a gradual, tunable increase in p‑type carrier concentration without introducing structural defects or external dopants.
The discussion highlights the practical relevance of these findings. Water and ammonia are abundant, environmentally benign species, suggesting that graphene devices could be doped in situ through simple exposure to controlled humid or ammonia atmospheres. Moreover, the reversible nature of hydrogen bonding implies that the doping level could be dynamically modulated by adjusting ambient conditions, opening pathways for sensor applications where the electronic response directly tracks molecular adsorption.
In conclusion, the study demonstrates that oxidized zigzag edges serve as an effective platform for the controlled p‑type doping of graphene via polar molecule adsorption. The combination of edge oxidation, hydrogen‑bond mediated charge transfer, and geometric tuning provides a versatile, defect‑free strategy for tailoring graphene’s electronic properties, with potential extensions to a broader class of polar or even non‑polar adsorbates, as well as to external stimuli such as electric fields or light.
Comments & Academic Discussion
Loading comments...
Leave a Comment