Dynamics of intracellular Ca$^{2+}$ oscillations in the presence of multisite Ca$^{2+}$-binding proteins
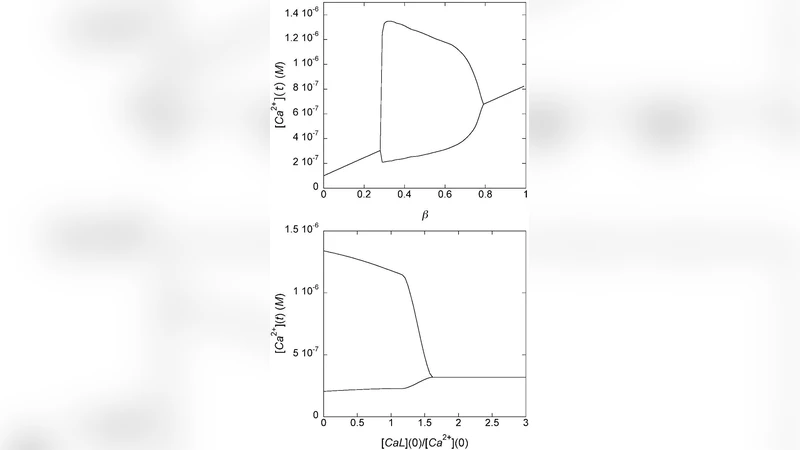
We study the dynamics of intracellular calcium oscillations in the presence of proteins that bind calcium on multiple sites and that are generally believed to act as passive calcium buffers in cells. We find that multisite calcium-binding proteins set a sharp threshold for calcium oscillations. Even with high concentrations of calcium-binding proteins, internal noise, which shows up spontaneously in cells in the process of calcium wave formation, can lead to self-oscillations. This produces oscillatory behaviors strikingly similar to those observed in real cells. In addition, for given intracellular concentrations of both calcium and calcium-binding proteins the regularity of these oscillations changes and reaches a maximum as a function noise variance, and the overall system dynamics displays stochastic coherence. We conclude that calcium-binding proteins may have an important and active role in cellular communication.
💡 Research Summary
The paper investigates how intracellular calcium (Ca²⁺) oscillations are shaped by proteins that possess multiple calcium‑binding sites, a class of molecules traditionally regarded as passive buffers. By extending the classic Li‑Rinzel model of calcium release and re‑uptake to explicitly include the binding‑unbinding kinetics of multisite proteins (e.g., calbindin‑D28k, parvalbumin), the authors construct a set of coupled differential equations that describe the full intracellular Ca²⁺ dynamics. Parameter sweeps reveal a sharp “oscillation‑threshold”: when the concentration of multisite buffers exceeds a critical value, the system settles into a stable fixed point and spontaneous Ca²⁺ spikes disappear. This demonstrates that such proteins do more than simply mop up free Ca²⁺; they actively reshape the nonlinear feedback loops that generate oscillations.
To capture the stochastic environment of real cells, the model incorporates internal noise as Gaussian white noise added to the calcium release term, representing random channel openings, molecular crowding, and other microscopic fluctuations. Remarkably, when the noise variance reaches an intermediate range, self‑sustained oscillations re‑emerge even below the deterministic threshold. The noise thus acts as a catalyst, pushing the system over a hidden bifurcation point. Moreover, the authors quantify the regularity of the resulting spikes by measuring the coefficient of variation of inter‑spike intervals. They find a non‑monotonic relationship: very low noise yields quiescence, very high noise produces irregular spikes, while a moderate noise level minimizes variability. This phenomenon, termed stochastic coherence, mirrors experimental observations of calcium signaling in neurons and muscle cells where the timing of spikes is surprisingly precise despite underlying molecular randomness.
Comparisons with experimental data show that cells rich in multisite buffers (e.g., certain inhibitory interneurons) often display suppressed calcium activity under basal conditions, yet can generate robust oscillations when intracellular noise is elevated (for instance, during high synaptic activity). Conversely, cells with low buffer concentrations exhibit oscillations even in the absence of significant noise. These findings suggest that multisite calcium‑binding proteins serve as tunable modulators of excitability, setting the sensitivity of the calcium signaling apparatus to both deterministic inputs and stochastic fluctuations.
In summary, the study overturns the simplistic view of multisite calcium‑binding proteins as passive sinks. It demonstrates that they impose a sharp threshold on calcium oscillations, that intrinsic cellular noise can overcome this threshold to produce self‑organized spikes, and that an optimal noise level yields maximal regularity—a clear case of stochastic coherence. Consequently, these proteins likely play an active, regulatory role in intracellular communication, influencing how cells encode and transmit information via calcium signals.
Comments & Academic Discussion
Loading comments...
Leave a Comment