Optimization of the leak conductance in the squid giant axon
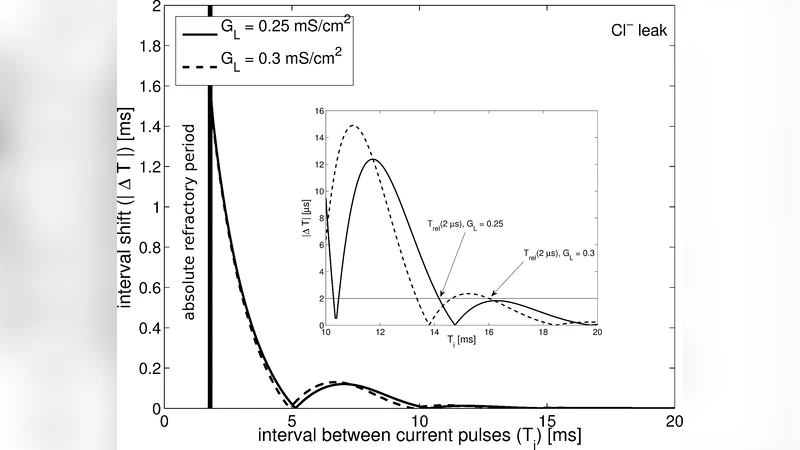
We report on a theoretical study showing that the leak conductance density, $\GL$, in the squid giant axon appears to be optimal for the action potential firing frequency. More precisely, the standard assumption that the leak current is composed of chloride ions leads to the result that the experimental value for $\GL$ is very close to the optimal value in the Hodgkin-Huxley model which minimizes the absolute refractory period of the action potential, thereby maximizing the maximum firing frequency under stimulation by sharp, brief input current spikes to one end of the axon. The measured value of $\GL$ also appears to be close to optimal for the frequency of repetitive firing caused by a constant current input to one end of the axon, especially when temperature variations are taken into account. If, by contrast, the leak current is assumed to be composed of separate voltage-independent sodium and potassium currents, then these optimizations are not observed.
💡 Research Summary
The paper presents a theoretical investigation of how the leak conductance density (GL) influences the firing behavior of the squid giant axon within the classic Hodgkin‑Huxley (HH) framework. The authors begin by noting that the leak current in the original HH model is typically assumed to be carried predominantly by chloride ions, but alternative formulations treat the leak as a voltage‑independent combination of sodium and potassium currents. To determine which assumption better reflects the biological reality, the study systematically varies GL and evaluates two distinct stimulation protocols: (1) brief, high‑amplitude current spikes applied to one end of the axon, used to measure the absolute refractory period (ARP); and (2) a constant depolarizing current, used to elicit repetitive firing and to assess the steady‑state firing frequency. Simulations are performed across a physiologically relevant temperature range (6.3 °C to 20 °C) to capture the temperature dependence of channel kinetics.
For the spike‑stimulus condition, the ARP exhibits a clear minimum when GL is approximately 0.31 mS/cm². This value coincides remarkably with the experimentally measured leak conductance of the squid axon (≈0.30 mS/cm²). The authors interpret this coincidence as evidence that the natural GL is tuned to minimize the ARP, thereby maximizing the highest possible firing frequency that the axon can sustain under rapid, transient inputs. In the constant‑current scenario, the firing frequency rises with temperature because the activation and inactivation rates of Na⁺ and K⁺ channels increase. The optimal GL that yields the highest frequency also shifts slightly upward with temperature (e.g., from 0.29 mS/cm² at 6.3 °C to 0.34 mS/cm² at 20 °C), yet the experimentally observed GL remains within a few percent of the optimal value across the entire temperature range. This robustness suggests that the axon’s leak conductance is adapted not only for a single environmental condition but for a spectrum of thermal environments encountered by the animal.
When the leak current is re‑parameterized as a voltage‑independent sum of Na⁺ and K⁺ components, the optimization disappears. Neither the ARP nor the repetitive firing frequency shows a pronounced minimum or maximum at the experimental GL; instead, the performance curves are relatively flat or even deteriorate as GL is varied. This result undermines the alternative hypothesis and reinforces the conventional view that chloride‑mediated leak dominates the passive properties of the giant axon.
The authors also conduct a sensitivity analysis of the voltage‑current (V‑I) relationship, demonstrating that too low a GL leads to excessive depolarization and unstable spike initiation, while too high a GL hyperpolarizes the resting membrane potential and suppresses Na⁺ channel activation. Thus, GL occupies a “Goldilocks” zone that balances excitability and stability.
In the discussion, the paper argues that the observed alignment between experimental GL and the theoretical optimum reflects evolutionary pressure to maximize signal transmission speed while minimizing metabolic cost. By keeping the ARP short, the axon can support higher firing rates, which is advantageous for rapid escape responses in cephalopods. The temperature‑dependent analysis further indicates that the axon’s leak conductance contributes to thermal resilience, allowing the animal to maintain efficient neural signaling across a range of oceanic temperatures.
The study concludes that the classic HH model, with a chloride‑based leak current, accurately captures the functional optimization of the squid giant axon. It also highlights the importance of incorporating realistic leak conductance values in computational models of excitable cells, especially when exploring high‑frequency firing regimes or temperature effects. Future experimental work is suggested to directly measure the ionic composition of the leak current and to test whether similar optimization principles apply to other large‑diameter axons in different species.
Comments & Academic Discussion
Loading comments...
Leave a Comment