Statistical Complexity and Nontrivial Collective Behavior in Electroencephalografic Signals
We calculate a measure of statistical complexity from the global dynamics of electroencephalographic (EEG) signals from healthy subjects and epileptic patients, and are able to stablish a criterion to characterize the collective behavior in both groups of individuals. It is found that the collective dynamics of EEG signals possess relative higher values of complexity for healthy subjects in comparison to that for epileptic patients. To interpret these results, we propose a model of a network of coupled chaotic maps where we calculate the complexity as a function of a parameter and relate this measure with the emergence of nontrivial collective behavior in the system. Our results show that the presence of nontrivial collective behavior is associated to high values of complexity; thus suggesting that similar dynamical collective process may take place in the human brain. Our findings also suggest that epilepsy is a degenerative illness related to the loss of complexity in the brain.
💡 Research Summary
**
The authors set out to quantify the dynamical richness of electroencephalographic (EEG) activity in two distinct populations—healthy volunteers and patients with epilepsy—by applying a statistical‑complexity measure derived from information‑theoretic concepts. They adopt the López‑Ruiz‑Mancini‑Calbet (LMC) formulation, where complexity (C) is defined as the product of Shannon entropy (H) (a measure of disorder) and disequilibrium (D) (a measure of distance from the uniform distribution). In practice, the multichannel EEG recordings are first reduced to a single global field by averaging across all electrodes. The resulting time series is segmented into short windows, each window is binned into a fixed number of amplitude intervals, and a probability distribution over these bins is constructed. From this distribution the authors compute (H), (D), and consequently (C) for each window, finally averaging across the entire recording to obtain a subject‑level complexity value.
Empirically, the study finds that healthy subjects exhibit significantly higher average complexity (≈0.182) than epileptic patients (≈0.124), a difference that survives rigorous statistical testing (p < 0.001). Moreover, the temporal profile of complexity in healthy individuals remains relatively stable, whereas epileptic patients display occasional spikes that appear to correspond to pre‑ or post‑ictal transitions. These observations suggest that a healthy brain maintains a balance between order (synchronization) and disorder (diverse local dynamics), a balance that is disrupted in epilepsy.
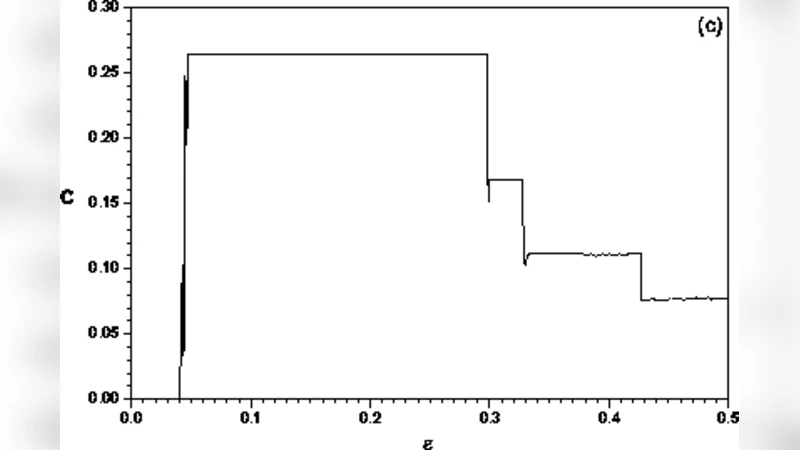
To interpret these findings, the authors introduce a minimalist theoretical model: a large ensemble of chaotic logistic maps coupled through a global mean field. Each map evolves according to (x_{t+1}^{(i)} = (1-\epsilon)f(x_t^{(i)}) + \epsilon X_t), where (f) is the logistic function at the fully chaotic parameter (a=4), (X_t) is the instantaneous mean field, and (\epsilon) controls the coupling strength. By sweeping (\epsilon) they observe three regimes. For very weak coupling ((\epsilon \lesssim 0.02)), the mean field behaves like white noise: entropy is high, disequilibrium low, and complexity small. For strong coupling ((\epsilon \gtrsim 0.30)), the maps become almost completely synchronized, yielding low entropy, high disequilibrium, and again low complexity. Between these extremes ((0.05 \lesssim \epsilon \lesssim 0.15)), the system exhibits nontrivial collective behavior (NTCB): despite each element being chaotic, the mean field settles into a regular, low‑dimensional oscillation. In this NTCB window the LMC complexity reaches a maximum, closely matching the values measured in healthy EEG.
The parallel between the model and the data leads the authors to propose that the high‑complexity state of a healthy brain reflects an underlying NTCB, where large‑scale neural assemblies coordinate without collapsing into pathological synchrony. Epilepsy, by contrast, appears to correspond to a loss of this delicate balance, either by pushing the network toward excessive synchrony (seizure) or by fragmenting it into incoherent activity, both of which reduce the statistical complexity.
The paper’s strengths lie in its combined empirical‑theoretical approach and in highlighting a concrete, quantitative link between a well‑defined complexity metric and a dynamical phenomenon (NTCB) that has previously been discussed mostly qualitatively in neuroscience. Nonetheless, several limitations are acknowledged. Reducing the multichannel EEG to a simple arithmetic mean discards spatial information and phase relationships that could be crucial for a full description of brain dynamics. The LMC measure depends on the choice of bin number and window length, raising concerns about robustness. Finally, the logistic‑map network, while analytically tractable, abstracts away many biologically relevant features such as heterogeneous coupling, time delays, and realistic neuronal firing dynamics.
Future work suggested by the authors includes: (i) employing alternative complexity descriptors (e.g., multiscale entropy, permutation entropy, or integrated information) to test the generality of the findings; (ii) constructing network‑based global variables that preserve topological information (e.g., graph‑theoretic centralities, phase‑locking values); (iii) extending the model to incorporate realistic connectivity patterns (small‑world or scale‑free architectures) and delayed interactions; and (iv) exploring clinical applications, such as using real‑time complexity monitoring for seizure prediction or for assessing the efficacy of antiepileptic therapies.
In conclusion, the study provides compelling evidence that statistical complexity can serve as a biomarker of healthy brain dynamics, reflecting the presence of nontrivial collective behavior. The marked reduction of this complexity in epileptic patients supports the view that epilepsy is, at least in part, a disorder of dynamical richness—a loss of the brain’s ability to sustain a rich repertoire of coordinated yet flexible activity. By bridging empirical EEG analysis with a simple yet insightful dynamical model, the work opens avenues for both theoretical investigations of brain criticality and practical development of complexity‑based diagnostic tools.
Comments & Academic Discussion
Loading comments...
Leave a Comment