Dynamics of path aggregation in the presence of turnover
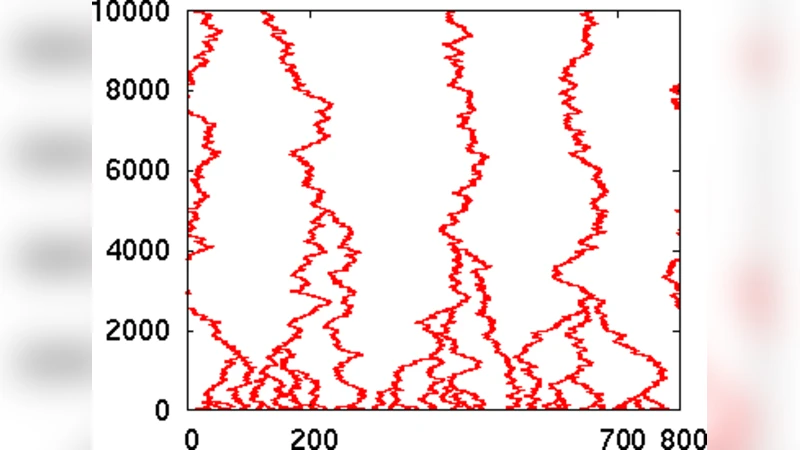
We investigate the slow time scales that arise from aging of the paths during the process of path aggregation. This is studied using Monte-Carlo simulations of a model aiming to describe the formation of fascicles of axons mediated by contact axon-axon interactions. The growing axons are represented as interacting directed random walks in two spatial dimensions. To mimic axonal turnover, random walkers are injected and whole paths of individual walkers are removed at specified rates. We identify several distinct time scales that emerge from the system dynamics and can exceed the average axonal lifetime by orders of magnitude. In the dynamical steady state, the position-dependent distribution of fascicle sizes obeys a scaling law. We discuss our findings in terms of an analytically tractable, effective model of fascicle dynamics.
💡 Research Summary
The paper addresses how axonal fasciculation— the bundling of growing axons into fascicles— evolves when axons are continuously turned over, i.e., newly generated and removed at a given rate. The authors model each axon as a directed random walk (DRW) on a two‑dimensional lattice that moves preferentially upward, mimicking axonal growth toward a target region. When two DRWs become adjacent, they interact and become part of the same fascicle; thereafter the whole fascicle moves as a single entity. To incorporate turnover, new walkers are injected at a constant rate λ_in while entire existing paths are removed after an average lifetime τ, representing axonal death and replacement.
Monte‑Carlo simulations were performed over a wide range of λ_in, τ, and system sizes. Several observables were recorded: the distribution of fascicle sizes P(s, x) as a function of the longitudinal coordinate x (distance from the origin), the number of fascicles N_f(x), the mean fascicle size ⟨s⟩(x), and temporal correlation functions that reveal characteristic relaxation times.
The results reveal three distinct dynamical time scales that can be orders of magnitude larger than the average axonal lifetime τ. First, the relaxation time of the mean fascicle size, τ_s, reflects how quickly the average bundle grows to its steady‑state value. Second, the fluctuation time of the fascicle number, τ_n, measures how long the system retains memory of a particular configuration of bundles. Third, a much longer “extinction” time τ_ex appears when a turnover event removes all existing paths simultaneously, causing a sudden collapse of the fascicle population. These time scales emerge from the collective dynamics of merging and splitting fascicles rather than from the individual walker lifetime.
In the steady state, the size distribution obeys a position‑dependent scaling law:
P(s, x) = x⁻¹ f(s/x)
where f is a universal, dimensionless scaling function that the simulations fit well with an exponential‑type form f(z) ∝ z exp(−z). This law implies that the average fascicle size grows linearly with distance (⟨s⟩ ∝ x) while the shape of the distribution remains invariant when sizes are rescaled by x. The scaling persists across a broad range of turnover rates, indicating that fascicle growth is self‑similar along the growth axis.
To rationalize these observations, the authors construct an analytically tractable “effective” model in which fascicles are treated as particles that undergo stochastic coalescence (merging) and fission (splitting). The coalescence rate depends on the product of the interacting fascicle sizes (∝√(s_i s_j)), reflecting the higher likelihood of larger bundles to attract each other. Splitting occurs with a rate proportional to fascicle size, representing internal instabilities that become more probable in larger bundles. Turnover introduces a global removal term with rate λ_out, which can trigger the extinction events described above. The master equation for the probability distribution of fascicle sizes can be solved in a mean‑field approximation, reproducing both the linear growth of ⟨s⟩ with x and the scaling form of P(s, x). Moreover, the analytical expressions for the characteristic times τ_s, τ_n, and τ_ex emerge naturally as functions of the coalescence, fission, and turnover rates.
The study highlights that fasciculation dynamics are governed by long‑range temporal correlations that far exceed the lifetime of individual axons. The presence of turnover does not simply randomize the system; instead, it creates a non‑trivial steady state where bundles continuously remodel while preserving a universal size distribution. These findings have broader implications for other biological systems where path‑like entities aggregate and turnover, such as vascular sprouting, insect trail formation, and collective robot navigation.
In conclusion, the authors provide a comprehensive computational and theoretical framework for understanding how axonal bundles form, evolve, and persist in the presence of continual renewal. By identifying multiple emergent time scales and a robust scaling law for fascicle sizes, the work bridges microscopic interaction rules with macroscopic tissue‑level organization, offering testable predictions for future experimental studies of neural development and related aggregation phenomena.
Comments & Academic Discussion
Loading comments...
Leave a Comment