Neuronal Shot Noise and Brownian $1/f^2$ Behavior in the Local Field Potential
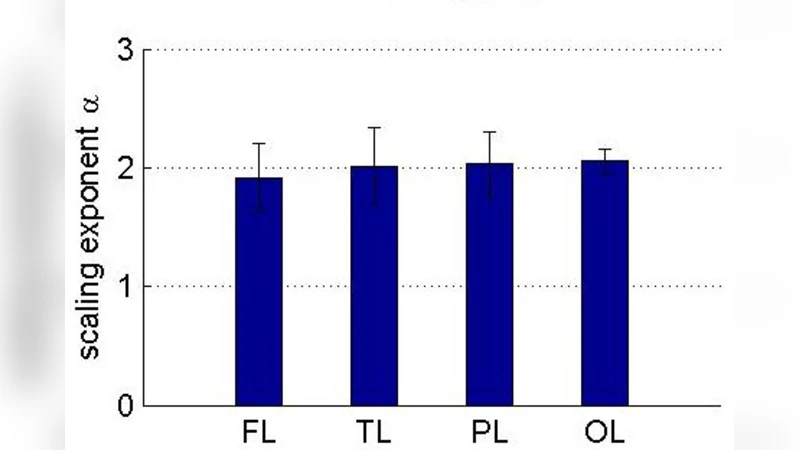
We demonstrate that human electrophysiological recordings of the local field potential (LFP) from intracranial electrodes, acquired from a variety of cerebral regions, show a ubiquitous $1/f^2$ scaling within the power spectrum. We develop a quantitative model that treats the generation of these fields in an analogous way to that of electronic shot noise, and use this model to specifically address the cause of this $1/f^2$ Brownian noise. The model gives way to two analytically tractable solutions, both displaying Brownian noise: 1) uncorrelated cells that display sharp initial activity, whose extracellular fields slowly decay and 2) rapidly firing, temporally correlated cells that generate UP-DOWN states.
💡 Research Summary
This paper reports a striking and pervasive 1/f² scaling in the power spectra of human intracranial local field potential (LFP) recordings obtained from a wide variety of cortical and subcortical regions. By analysing data from multiple patients and electrode sites, the authors demonstrate that across a broad frequency band (approximately 1 Hz to several hundred Hz) the spectral density follows a near‑perfect −2 slope on log–log axes, indicating Brownian‑type noise rather than the more commonly reported 1/f or 1/f³ scaling.
To explain this phenomenon, the authors construct a quantitative model that treats each neuronal discharge as an elementary “charge packet” analogous to electronic shot noise. In the classic shot‑noise picture, discrete charge carriers are emitted according to a Poisson process; the resulting current consists of delta‑function spikes that, when passed through a low‑pass filter (or integrated over time), acquire a 1/f² power spectrum. Translating this to neurophysiology, each neuron’s action potential generates a rapid extracellular current pulse, which then decays according to a kernel K(t) that captures the biophysical spread of the field through the surrounding tissue. The recorded LFP is the linear superposition of N such events:
LFP(t) = Σ_i ∫ δ(t – t_i) K(t – τ) dτ,
where t_i are the spike times of neuron i. The model therefore reduces the problem to two key ingredients: (1) the statistical distribution of spike times (Poisson‑like or correlated) and (2) the temporal shape of the decay kernel.
Two analytically tractable regimes are explored.
-
Uncorrelated neurons with sharp spikes – Here each neuron fires independently, producing an almost instantaneous current pulse. If the decay kernel follows a 1/t or slowly exponential form, the integral of the Poisson process yields a power spectrum S(f) ∝ 1/f². This regime corresponds to a classic shot‑noise scenario: many independent “shots” whose cumulative effect is Brownian noise.
-
Rapidly firing, temporally correlated neurons (UP‑DOWN states) – In many cortical circuits, neurons participate in network‑wide oscillations that alternate between high‑activity (UP) and low‑activity (DOWN) epochs. The spike train is no longer a simple Poisson process but exhibits long‑range temporal correlations, often modeled by an autocorrelation function C(τ) ∝ e^{‑λ|τ|}. Incorporating this correlation into the shot‑noise framework still leads to S(f) ∝ 1/f², because the low‑frequency power is dominated by the slow switching between states. This solution captures the contribution of coordinated network dynamics to the observed Brownian spectrum.
Both solutions reproduce the experimentally observed 1/f² scaling, suggesting that the LFP’s spectral shape can arise from either a multitude of independent, sharply timed events or from a smaller set of highly synchronized, slowly switching ensembles. The authors validate the model by comparing simulated spectra with the recorded data, showing close agreement in slope, amplitude, and robustness across electrode locations and brain regions.
The paper’s contributions are threefold. First, it provides a unified physical‑mathematical framework that links neuronal field generation to the well‑understood phenomenon of electronic shot noise, thereby offering a fresh perspective on why LFPs often display Brownian characteristics. Second, it identifies two distinct physiological mechanisms—uncorrelated “shot” firing and correlated UP‑DOWN dynamics—that can each generate the same spectral signature, implying that real brain activity likely reflects a mixture of both. Third, the model’s analytical tractability makes it a promising tool for interpreting alterations in LFP spectra observed in neurological disorders (e.g., epilepsy, Parkinson’s disease) where either the independence of neuronal firing or the structure of network states is disrupted.
Nevertheless, the model simplifies several important aspects of brain electrophysiology. It assumes linear superposition of fields, neglects the anisotropic and heterogeneous conductivity of brain tissue, and treats the decay kernel with a fixed functional form. Synaptic currents, active dendritic processes, and non‑linear membrane dynamics are not explicitly modeled, which could modify the high‑frequency portion of the spectrum. Moreover, the clinical recordings used in the study are subject to electrode geometry, placement constraints, and environmental noise, factors that may bias the observed scaling.
Future work should aim to (i) record simultaneously from dense micro‑electrode arrays to directly capture the spike‑field relationship at the single‑cell level, (ii) incorporate realistic volume‑conductor models that account for tissue inhomogeneities, and (iii) extend the framework to include non‑linear source dynamics and state‑dependent conductances. Such extensions would test the robustness of the 1/f² prediction and clarify how pathological changes in neuronal synchrony or firing statistics manifest in the LFP power spectrum.
In summary, the authors convincingly demonstrate that the ubiquitous 1/f² scaling of human LFPs can be understood as a form of neuronal shot noise, with both uncorrelated rapid spikes and correlated UP‑DOWN network states providing viable mechanistic explanations. This work bridges concepts from electronic noise theory and neurophysiology, offering a powerful analytical lens for future investigations of brain dynamics and their alterations in disease.
Comments & Academic Discussion
Loading comments...
Leave a Comment