Regulation of signal duration and the statistical dynamics of kinase activation by scaffold proteins
Scaffolding proteins that direct the assembly of multiple kinases into a spatially localized signaling complex are often essential for the maintenance of an appropriate biological response. Although scaffolds are widely believed to have dramatic effects on the dynamics of signal propagation, the mechanisms that underlie these consequences are not well understood. Here, Monte Carlo simulations of a model kinase cascade are used to investigate how the temporal characteristics of signaling cascades can be influenced by the presence of scaffold proteins. Specifically, we examine the effects of spatially localizing kinase components on a scaffold on signaling dynamics. The simulations indicate that a major effect that scaffolds exert on the dynamics of cell signaling is to control how the activation of protein kinases is distributed over time. Scaffolds can influence the timing of kinase activation by allowing for kinases to become activated over a broad range of times, thus allowing for signaling at both early and late times. Scaffold concentrations that result in optimal signal amplitude also result in the broadest distributions of times over which kinases are activated. These calculations provide insights into one mechanism that describes how the duration of a signal can potentially be regulated in a scaffold mediated protein kinase cascade. Our results illustrate another complexity in the broad array of control properties that emerge from the physical effects of spatially localizing components of kinase cascades on scaffold proteins.
💡 Research Summary
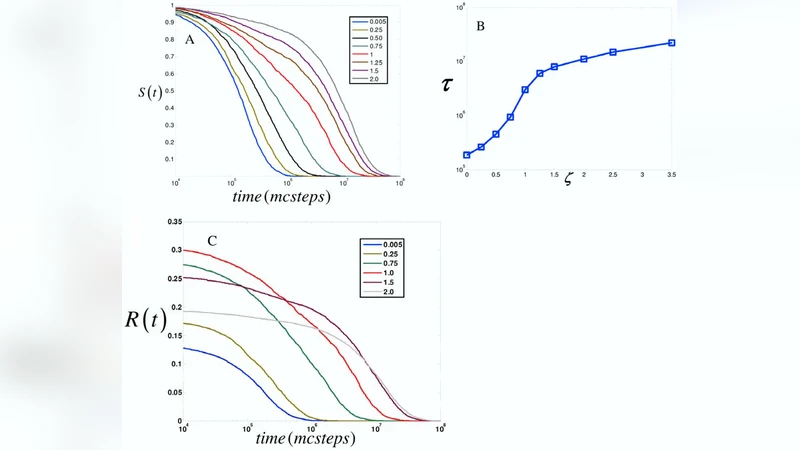
The paper investigates how scaffold proteins shape the temporal dynamics of kinase cascades by using Monte Carlo simulations of a three‑step MAPK pathway. The authors construct a stochastic model in which each kinase can exist either freely in the cytosol or bound to a scaffold that brings the three kinases into close proximity. Key parameters include the scaffold concentration, the on‑ and off‑rates for each kinase–scaffold interaction, and the catalytic rates for activation and deactivation. By systematically varying scaffold concentration from zero to saturating levels, the simulations generate thousands of individual molecular trajectories that allow the authors to extract statistical properties of the signaling response, such as peak amplitude, time‑to‑peak, and the distribution of activation times across the population of MAPK molecules.
The results reveal a non‑monotonic relationship between scaffold concentration and signal characteristics. At low scaffold levels, most kinases remain free; activation occurs quickly but the signal decays rapidly, producing a narrow, early‑time peak. At very high scaffold concentrations, the majority of kinases become sequestered on scaffolds, which limits their ability to encounter substrates and leads to a reduced overall amplitude. In an intermediate “optimal” range of scaffold concentration, both free and scaffold‑bound pathways contribute. The scaffold accelerates early activation by increasing effective collision frequency, while the gradual dissociation of scaffold‑kinase complexes sustains activation at later times. Consequently, the distribution of activation times becomes the broadest, and the signal persists for a much longer duration. Notably, the scaffold concentration that maximizes signal amplitude also yields the widest temporal distribution, indicating that scaffolds simultaneously regulate signal strength and duration.
The authors interpret these findings as evidence that scaffolds act as temporal filters: they can spread kinase activation over a wide time window, allowing a single stimulus to generate both rapid and prolonged downstream responses. The physical trade‑off underlying this behavior is the balance between enhanced proximity (which speeds up enzymatic steps) and excessive sequestration (which hampers diffusion‑limited encounters). By adjusting binding affinities and off‑rates, cells can fine‑tune this balance to meet specific physiological demands.
Finally, the paper suggests experimental strategies to validate the model, such as titrating scaffold expression or introducing point mutations that alter kinase‑scaffold affinity, followed by quantitative live‑cell imaging of MAPK activation dynamics. The authors also discuss implications for disease contexts where scaffold function is perturbed, for example in cancers with aberrant MAPK signaling. Overall, the study provides a quantitative framework that links the spatial organization imposed by scaffold proteins to the statistical dynamics of signal propagation, highlighting scaffold‑mediated control of signal duration as a fundamental principle of cellular information processing.
Comments & Academic Discussion
Loading comments...
Leave a Comment