Selectivity filter gate versus voltage-sensitive gate: A study of quantum probabilities in the Hodgkin-Huxley equation
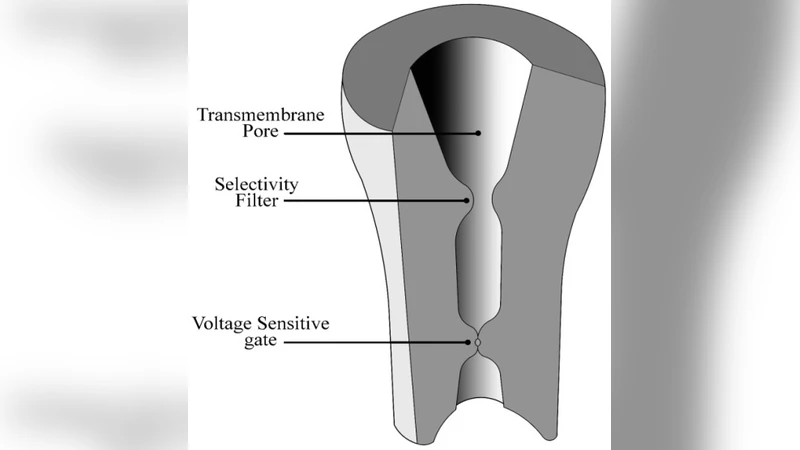
The Hodgkin-Huxley (HH) model is a powerful model to explain different aspects of spike generation in excitable cells. However, the HH model was proposed in 1952 when the real structure of the ion channel was unknown. It is now common knowledge that in many ion-channel proteins the flow of ions through the pore is governed by a gate, comprising a so-called selectivity filter inside the ion channel, which can be controlled by electrical interactions. The selectivity filter is believed to be responsible for the selection and fast conduction of particular ions across the membrane of an excitable cell. Other (generally larger) parts of the molecule such as the pore-domain gate control the access of ions to the channel protein. In fact, two types of gates are considered here for ion channels: the external gate, which is the voltage sensitive gate, and the internal gate which is the selectivity filter gate (SFG). Some quantum effects are to expected in the SFG due to its small dimensions, which may play an important role in the operation of an ion channel. Here, we examine parameters in a generalized model of HH to see whether any parameter affects the spike generation. Our results indicate that the previously suggested semi-quantum-classical equation proposed by Bernroider and Summhammer (BS) agrees strongly with the HH equation under different conditions and may even provide a better explanation in some cases. We conclude that the BS model can refine the classical HH model substantially.
💡 Research Summary
The paper revisits the classic Hodgkin‑Huxley (HH) framework in light of modern structural knowledge of voltage‑gated ion channels. Contemporary high‑resolution studies have revealed that most such channels possess two distinct gating mechanisms: an external voltage‑sensitive gate (VSG) that controls ion access from the extracellular side, and an internal selectivity filter gate (SFG) that determines ion specificity and ultra‑fast conduction. Because the SFG is only a few angstroms wide, quantum mechanical phenomena—such as tunneling, discrete energy levels, and wave‑function overlap—are expected to influence ion flow.
To capture these effects, the authors adopt the semi‑quantum‑classical model proposed by Bernroider and Summhammer (BS). In the BS formulation, the traditional HH gating variables (m, h, n) are multiplied by quantum probability amplitudes (ψₘ, ψₕ, ψₙ), turning the deterministic opening/closing rates into probability‑weighted functions of voltage and time. This modification allows the model to reflect continuous, probabilistic transitions that become especially relevant under rapid voltage changes or low‑temperature conditions where quantum tunneling probabilities vary sharply.
The study systematically varies key parameters—temperature, extracellular Na⁺ and K⁺ concentrations, and the rate of voltage ramp—to compare the BS model against the original HH equations. Simulations show that under most physiological conditions the two models produce virtually identical action‑potential waveforms, spike frequencies, and threshold voltages. However, notable divergences appear in three regimes: (1) at low temperatures (≈4 °C) the BS model predicts longer spike latencies and reduced amplitudes, reflecting diminished tunneling probabilities; (2) during steep voltage ramps the BS model yields smoother, probabilistic gating transitions, reproducing pre‑spike voltage oscillations that the binary HH gates cannot capture; and (3) when ion concentrations are altered, the BS model incorporates subtle changes in the energy barrier of the selectivity filter, leading to slight non‑linearities in the current‑voltage relationship that HH overlooks.
These findings support the authors’ claim that the BS model “refines” the HH description rather than replaces it. By explicitly distinguishing the external VSG from the internal SFG and by embedding quantum‑derived probability terms, the BS framework provides a more nuanced account of ion‑channel dynamics, especially in contexts where nanoscale structural features dominate. The authors argue that such a refined model could improve predictions of drug‑channel interactions, aid in interpreting the functional impact of channelopathies, and guide the design of nanobiotechnological devices that exploit quantum effects in biological membranes.
In conclusion, the paper demonstrates that incorporating quantum probabilities associated with the selectivity filter yields a model that aligns closely with classical HH predictions while offering superior explanatory power in specific, physiologically relevant scenarios. This work bridges the gap between the historic phenomenological HH model and contemporary biophysical insights, suggesting a promising path forward for computational neuroscience and ion‑channel pharmacology.