Entrainment of heterogeneous glycolytic oscillations in single cells
Cell signaling, gene expression, and metabolism are affected by cell-cell heterogeneity and random changes in the environment. The effects of such fluctuations on cell signaling and gene expression have recently been studied intensively using single-cell experiments. In metabolism heterogeneity may be particularly important because it may affect synchronisation of metabolic oscillations, an important example of cell-cell communication. This synchronisation is notoriously difficult to describe theoretically as the example of glycolytic oscillations shows: neither is the mechanism of glycolytic synchronisation understood nor the role of cell-cell heterogeneity. To pin down the mechanism and to assess its robustness and universality we have experimentally investigated the entrainment of glycolytic oscillations in individual yeast cells by periodic external perturbations. We find that oscillatory cells synchronise through phase shifts and that the mechanism is insensitive to cell heterogeneity (robustness) and similar for different types of external perturbations (universality).
💡 Research Summary
This paper investigates how heterogeneous glycolytic oscillations in individual yeast cells can be entrained by periodic external perturbations, addressing a long‑standing problem in metabolic synchronization. The authors use single‑cell fluorescence imaging of NADH to monitor glycolytic oscillations in Saccharomyces cerevisiae confined within microfluidic chambers. Two distinct perturbation modalities are applied: (i) brief glucose concentration pulses (±0.2 mM) and (ii) short pH pulses (±0.05 pH units). Both perturbations are delivered with pulse durations of 0.5–2 seconds and with forcing periods ranging from 30 s to 120 s.
For each cell, the instantaneous phase of the oscillation is extracted via a Hilbert transform, allowing the construction of phase‑response curves (PRCs). The PRCs reveal a characteristic type‑1 shape: perturbations delivered during the rising phase of the NADH signal produce large phase advances, whereas those applied during the falling phase cause only modest delays. This asymmetry indicates that the external rhythm can reset the oscillator’s phase efficiently when timed appropriately.
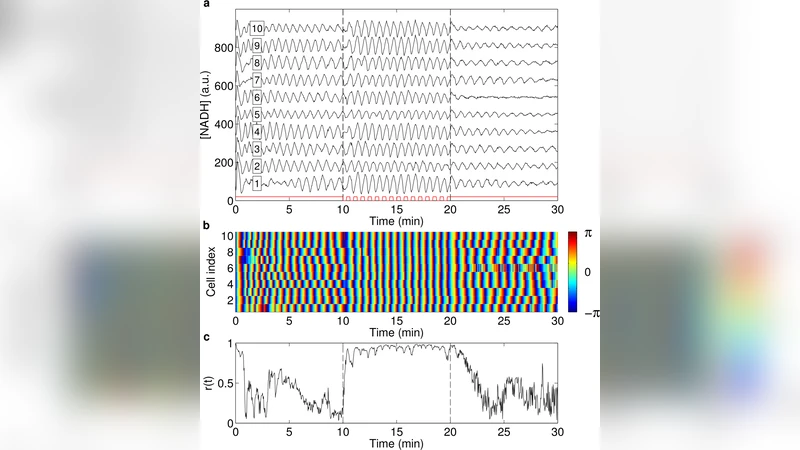
Prior to forcing, the population exhibits substantial heterogeneity: the intrinsic periods are normally distributed around 45 s with a standard deviation of ~12 s, and amplitudes vary by roughly 30 %. Despite this variability, after the onset of periodic forcing the order parameter (a measure of collective phase coherence) jumps from ~0.2 to >0.9, indicating near‑perfect synchronization. The synchronization is strongest when the forcing period is close to the mean intrinsic period, reproducing the classic Arnold‑tongue behavior predicted by forced oscillator theory.
Importantly, both glucose and pH perturbations generate virtually identical PRCs and achieve comparable synchronization efficiencies, demonstrating a form of universality: the entrainment mechanism does not depend on the chemical nature of the stimulus but rather on its ability to induce a rapid, transient shift in the metabolic state. To rationalize these observations, the authors extend the Kuramoto model by incorporating a nonlinear PRC and explicit heterogeneity parameters. Numerical simulations of this extended model reproduce the experimental PRCs, the rapid rise in the order parameter, and the shape of the Arnold‑tongue region, confirming that phase resetting is the dominant entrainment mechanism.
The study concludes that glycolytic oscillations are robustly entrainable through phase shifts induced by brief external cues, and that this mechanism tolerates substantial cell‑to‑cell variability. The findings have broad implications: they suggest that metabolic networks can synchronize across heterogeneous populations without requiring precise coupling, and they provide a quantitative framework that may be applicable to other biochemical oscillators such as calcium or cAMP rhythms. The authors propose future work to explore spatial propagation of entrainment in multicellular tissues and to examine how pathological metabolic reprogramming (e.g., in cancer) might alter the entrainment dynamics.
Comments & Academic Discussion
Loading comments...
Leave a Comment