Investigation of bone resorption within a cortical basic multicellular unit using a lattice-based computational model
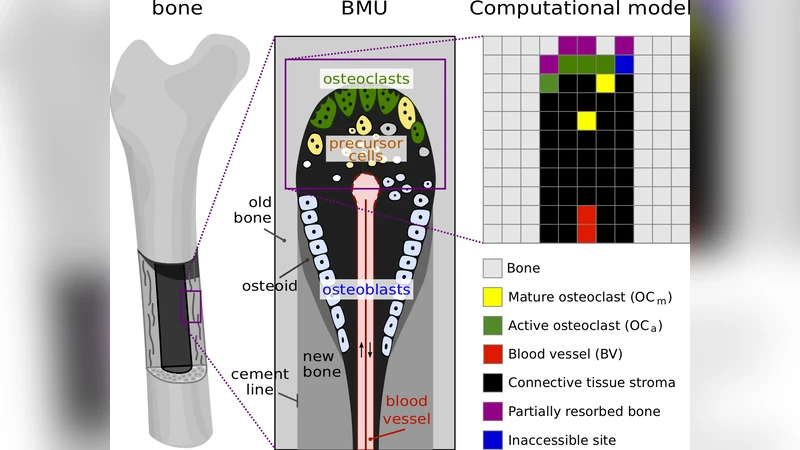
In this paper we develop a lattice-based computational model focused on bone resorption by osteoclasts in a single cortical basic multicellular unit (BMU). Our model takes into account the interaction of osteoclasts with the bone matrix, the interaction of osteoclasts with each other, the generation of osteoclasts from a growing blood vessel, and the renewal of osteoclast nuclei by cell fusion. All these features are shown to strongly influence the geometrical properties of the developing resorption cavity including its size, shape and progression rate, and are also shown to influence the distribution, resorption pattern and trajectories of individual osteoclasts within the BMU. We demonstrate that for certain parameter combinations, resorption cavity shapes can be recovered from the computational model that closely resemble resorption cavity shapes observed from microCT imaging of human cortical bone.
💡 Research Summary
This paper presents a lattice‑based, agent‑based computational model that simulates bone resorption by osteoclasts within a single cortical basic multicellular unit (BMU). The authors discretize a two‑dimensional cross‑section of cortical bone into a square lattice with a spacing of 40 µm, which corresponds to the average size of a multinucleated osteoclast. Each lattice site can represent bone, partially resorbed bone, fully resorbed cavity (filled with connective tissue), an active resorbing osteoclast (OC_a), a migrating osteoclast (OC_m), blood‑vessel components, or stromal tissue.
Key biological processes incorporated are: (1) generation of mature osteoclasts at a rate η_OC from the tip of a blood vessel that itself advances forward at a maximal speed v_BV; (2) a binary state for each osteoclast – migrating (OC_m) or actively resorbing (OC_a). Upon contact with bone, an OC_m instantly becomes OC_a, remains immobile while it dissolves surrounding bone, and reverts to OC_m once all adjacent bone has been removed; (3) bone dissolution follows a kinetic law that gradually reduces local bone density, and sites that reach >90 % resorption become inaccessible connective‑tissue cavities for a refractory period τ_inhib, mimicking extracellular collagen digestion; (4) migration of OC_m is modeled as a biased random walk driven by interaction energies (e.g., osteoclast‑osteoclast adhesion E_OC‑OC, osteoclast‑bone adhesion E_OC‑BONE). The probability of moving to a neighboring site is higher when the total interaction energy is lower; (5) each newly created osteoclast receives a fixed lifespan τ_OC, but this lifespan can be extended through nuclear fusion events. A migrating OC_m may fuse with an existing OC_a or another OC_m, with fusion probabilities governed by fusion energies (E_fusion). The resulting cell inherits the sum of the remaining lifespans of the fusing partners, reflecting experimentally observed thymidine‑labelled nuclei renewal.
Through systematic parameter sweeps the authors identify how each component shapes the emerging resorption cavity (cutting cone). Strong osteoclast‑osteoclast adhesion leads to clustering, preventing uniform bone access and producing abnormally narrow cones, whereas weak adhesion yields dispersed cells that cover the surface but slow overall progression. An optimal osteoclast‑bone adhesion is required to balance stable attachment with the ability to detach and relocate, generating smooth, ellipsoidal cavities. The spatial position of the blood‑vessel tip is critical: when the tip lies near the leading edge of the cavity, a steady supply of new osteoclasts sustains symmetric growth; if the tip lags behind, cell supply becomes limiting and the cavity becomes asymmetric. Nuclear fusion and the associated lifespan extension enable osteoclasts to remain active long enough for the cutting cone to advance at a rate comparable to the vessel growth speed, thereby coupling cellular turnover to BMU progression.
A specific set of parameters (e.g., η_OC≈0.5 day⁻¹, v_BV≈30 µm day⁻¹, E_OC‑OC≈‑2 k_BT, E_OC‑BONE≈‑5 k_BT, τ_inhib≈2 days, τ_OC≈10 days, E_fusion≈‑3 k_BT) reproduces cutting‑cone morphologies that closely match those observed in human cortical bone micro‑CT scans: ellipsoidal cross‑sections with a characteristic rough surface. The model also predicts osteoclast migration trajectories, temporary detachment‑reattachment cycles, and the spatial distribution of nuclear renewal—phenomena that are currently difficult to capture experimentally.
In the discussion, the authors contrast their discrete, cell‑level approach with earlier continuum models that rely on averaged cell densities and mechanical cues alone. By explicitly representing cell‑cell and cell‑matrix adhesion, blood‑vessel driven cell supply, and nuclear fusion, the model provides mechanistic insight into how microscopic cellular behaviors translate into macroscopic BMU geometry and progression speed. Limitations are acknowledged: the model excludes osteoblasts, osteocytes, endothelial signaling, and explicit biochemical pathways; it also operates in two dimensions. Future work is proposed to extend the framework to three dimensions, incorporate additional cell types and signaling networks, and couple the resorption model with mechanical loading to explore the interplay between biomechanics and cellular dynamics in bone remodeling.
Comments & Academic Discussion
Loading comments...
Leave a Comment