Rate Control Management of Atrial Fibrillation: May a Mathematical Model Suggest an Ideal Heart Rate?
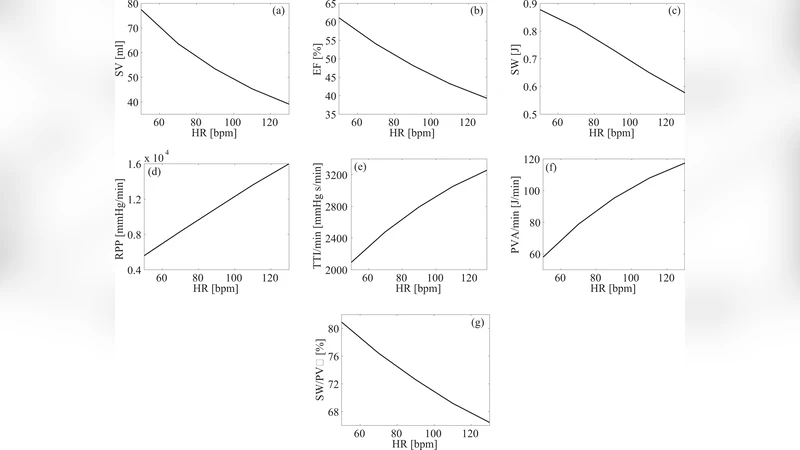
Background. Despite the routine prescription of rate control therapy for atrial fibrillation (AF), clinical evidence demonstrating a heart rate target is lacking. Aim of the present study was to run a mathematical model simulating AF episodes with a different heart rate (HR) to predict hemodynamic parameters for each situation. Methods. The lumped model, representing the pumping heart together with systemic and pulmonary circuits, was run to simulate AF with HR of 50, 70, 90, 110 and 130 bpm, respectively. Results. Left ventricular pressure increased by 56.7%, from 33.92+-37.56 mmHg to 53.15+-47.56 mmHg, and mean systemic arterial pressure increased by 27.4%, from 82.66+-14.04 mmHg to 105.29+-7.63 mmHg, at the 50 and 130 bpm simulations, respectively. Stroke volume (from 77.45+-8.5 to 39.09+-8.08 mL), ejection fraction (from 61.1+-4.4 to 39.32+-5.42%) and stroke work (SW, from 0.88+-0.04 to 0.58+-0.09 J) decreased by 49.5, 35.6 and 34.2%, at the 50 and 130 bpm simulations, respectively. In addition, oxygen consumption indexes (rate pressure product, RPP, tension time index per minute, TTI/min, and pressure volume area per minute, PVA/min) increased from the 50 to the 130 bpm simulation, respectively, by 185.7% (from 5598+-1939 to 15995+-3219 mmHg/min), 55.5% (from 2094+-265 to 3257+-301 mmHg s/min) and 102.4% (from 57.99+-17.9 to 117.37+-25.96 J/min). In fact, left ventricular efficiency (SW/PVA) decreased from 80.91+-2.91% at 50 bpm to 66.43+-3.72% at the 130 bpm HR simulation. Conclusion. Awaiting compulsory direct clinical evidences, the present mathematical model suggests that lower HRs during permanent AF relates to improved hemodynamic parameters, cardiac efficiency, and lower oxygen consumption.
💡 Research Summary
The authors address a persistent gap in atrial fibrillation (AF) management: the lack of solid evidence for an optimal heart‑rate (HR) target when employing rate‑control therapy. To explore this, they constructed a zero‑dimensional (lumped‑parameter) cardiovascular model that represents the left and right ventricles, systemic and pulmonary circuits, and the major arterial and venous segments as resistors, capacitors, and inductors. The model reproduces the pressure‑flow dynamics of the entire circulation with a set of first‑order differential equations.
AF was simulated by removing atrial contribution to ventricular filling and by imposing a stochastic sequence of RR intervals drawn from a normal distribution whose mean equals 60 seconds divided by the chosen HR. Ventricular contractility (time‑varying elastance) was held constant across simulations, reflecting a “typical” healthy myocardium. Five HR scenarios were examined: 50, 70, 90, 110, and 130 beats per minute (bpm). Each simulation ran for 60 seconds and was repeated ten times to obtain mean values and standard deviations for the following variables: left‑ventricular systolic pressure (LVPs), mean arterial pressure (MAP), stroke volume (SV), ejection fraction (EF), stroke work (SW), pressure‑volume area (PVA), and three surrogate indices of myocardial oxygen consumption—rate‑pressure product (RPP), tension‑time index per minute (TTI/min), and PVA per minute. Cardiac efficiency was expressed as the ratio SW/PVA.
The results reveal a clear, monotonic relationship between HR and the hemodynamic and metabolic outcomes. As HR increased from 50 to 130 bpm, LVPs rose from 33.9 ± 37.6 mmHg to 53.2 ± 47.6 mmHg (≈57 % increase) and MAP rose from 82.7 ± 14.0 mmHg to 105.3 ± 7.6 mmHg (≈27 % increase). In contrast, SV fell dramatically from 77.5 ± 8.5 mL to 39.1 ± 8.1 mL (≈49 % reduction) and EF dropped from 61.1 ± 4.4 % to 39.3 ± 5.4 % (≈36 % reduction). Stroke work decreased from 0.88 ± 0.04 J to 0.58 ± 0.09 J (≈34 % reduction), while PVA increased from 0.69 J to 0.87 J (≈26 % increase). Consequently, cardiac efficiency (SW/PVA) declined from 80.9 ± 2.9 % at 50 bpm to 66.4 ± 3.7 % at 130 bpm.
Metabolic demand rose sharply with HR. RPP, a classic index of myocardial oxygen consumption, more than doubled, climbing from 5,598 ± 1,939 mmHg·bpm at 50 bpm to 15,995 ± 3,219 mmHg·bpm at 130 bpm (≈186 % increase). TTI/min grew by 55 % (2,094 ± 265 → 3,257 ± 301 mmHg·s/min) and PVA/min more than doubled (57.99 ± 17.9 J/min → 117.37 ± 25.96 J/min, ≈102 % increase).
These findings support the hypothesis that, in permanent AF, a lower HR yields a more favorable hemodynamic profile: higher systolic pressures are offset by preserved stroke volume and ejection fraction, resulting in greater cardiac efficiency and markedly lower myocardial oxygen consumption. The model therefore provides a quantitative rationale for the current guideline recommendation to aim for a “slow” ventricular response (generally ≤80 bpm, preferably ≤70 bpm) in AF patients.
Nevertheless, the study has several limitations that temper the direct clinical translatability of the results. First, ventricular contractility was assumed constant, whereas in real‑world AF patients myocardial inotropy may be depressed by remodeling, ischemia, or pharmacologic agents. Second, the vascular beds were represented by linear resistors and capacitors, ignoring the non‑linear compliance, autoregulatory mechanisms, and neuro‑humoral influences that modulate afterload and preload in vivo. Third, the simulation assumes complete loss of atrial contribution, yet many patients retain some atrial mechanical activity or experience varying degrees of atrioventricular synchrony. Fourth, the parameter set reflects an average healthy adult; extrapolation to elderly, hypertensive, or heart‑failure populations would require recalibration.
In summary, the paper demonstrates that a lumped‑parameter cardiovascular model can be a powerful tool for exploring the physiological consequences of different rate‑control strategies in AF. By systematically varying HR, the authors show that lower ventricular rates improve stroke volume, ejection fraction, and overall cardiac efficiency while reducing indices of myocardial oxygen demand. These insights reinforce the clinical practice of targeting a relatively slow heart rate in permanent AF, while also highlighting the need for patient‑specific modeling and prospective clinical validation before the findings can be fully integrated into therapeutic decision‑making.
Comments & Academic Discussion
Loading comments...
Leave a Comment