Predicting rare events in chemical reactions: application to skin cell proliferation
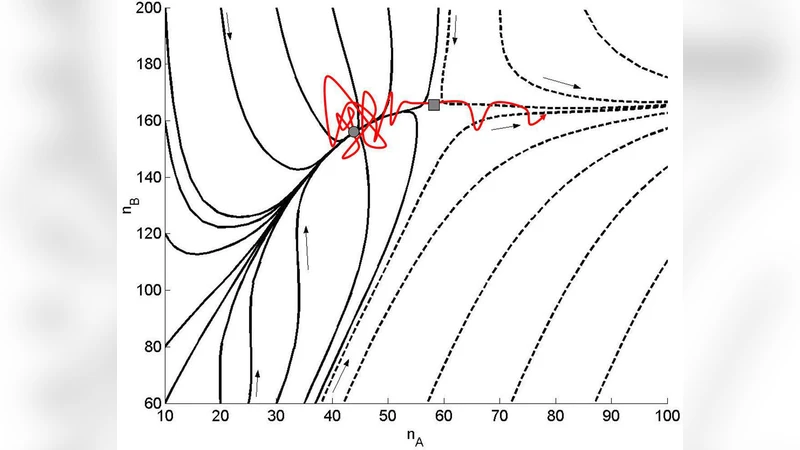
In a well-stirred system undergoing chemical reactions, fluctuations in the reaction propensities are approximately captured by the corresponding chemical Langevin equation. Within this context, we discuss in this work how the Kramers escape theory can be used to predict rare events in chemical reactions. As an example, we apply our approach to a recently proposed model on cell proliferation with relevance to skin cancer [P.B. Warren, Phys. Rev. E {\bf 80}, 030903 (2009)]. In particular, we provide an analytical explanation for the form of the exponential exponent observed in the onset rate of uncontrolled cell proliferation.
💡 Research Summary
The paper presents a theoretical framework for predicting rare events in stochastic chemical reaction systems by combining the chemical Langevin equation (CLE) with Kramers escape theory. The authors begin by noting that in a well‑stirred reactor the discrete stochastic dynamics described by the master equation can be approximated, for sufficiently large molecule numbers, by a continuous stochastic differential equation – the CLE. This equation captures both the deterministic drift given by the law of mass action and a diffusion term that represents intrinsic noise arising from the discreteness of reaction events.
Kramers theory, originally developed to describe thermally activated escape over an energy barrier, provides an expression for the escape rate ( r \approx A \exp(-\Delta U/D) ), where ( \Delta U ) is the barrier height and ( D ) is the noise intensity. By mapping the state space of a chemical reaction network onto an effective potential landscape, the authors treat the transition from a stable fixed point to another (often unstable) region as a barrier‑crossing problem. The key technical step is to identify the most probable transition path – the minimum‑action trajectory – using a variational principle applied to the CLE‑derived action functional. The action evaluated along this optimal path, denoted ( S^* ), determines the exponential factor of the escape rate: ( r \approx C \exp(-S^*/\varepsilon) ), where ( \varepsilon ) is a small parameter inversely proportional to system size and ( C ) is a prefactor that depends on curvature of the effective potential at the fixed points.
To demonstrate the utility of this approach, the authors apply it to a recently proposed model of skin cell proliferation (Warren, Phys. Rev. E 80, 030903, 2009). The model distinguishes between normal keratinocytes (N) and mutant cells (M) and includes stochastic reactions for proliferation, death, and conversion between the two types. In the deterministic limit the system possesses two fixed points: a stable “normal” state and an unstable “runaway” state where mutant cells proliferate without bound. The stochastic dynamics allow the system to escape from the normal basin and enter the runaway basin, an event that corresponds to the onset of uncontrolled cell growth – a rare but biologically critical transition.
The authors first derive the mean‑field equations, locate the fixed points, and then construct the CLE for the two‑dimensional system. By projecting the dynamics onto the (N, M) plane, they compute the optimal escape trajectory using a Hamiltonian formulation of the action. Numerical evaluation of the action yields a simple logarithmic dependence on the ratio of mutant proliferation to death rates, ( \lambda/\mu ). Specifically, the exponent takes the form ( S^* \approx \alpha \ln(\lambda/\mu) ), leading to an escape rate ( r \sim (\mu/\lambda)^{\alpha} ). This reproduces the exponential suppression observed in Warren’s simulations and provides an analytical explanation for the empirical exponent.
The prefactor ( C ) is shown to scale with the initial population of normal cells and with the probability of a normal cell converting into a mutant. Consequently, larger initial clones or higher conversion probabilities increase the likelihood of a rare runaway event, consistent with epidemiological observations that larger patches of mutated skin carry higher cancer risk.
Beyond this specific biological example, the paper argues that the CLE + Kramers framework is broadly applicable to any multistable chemical or biochemical network where rare transitions dominate system behavior. The methodology offers a systematic way to compute both the exponential barrier term and the sub‑exponential prefactor, enabling quantitative risk assessment for phenomena such as nucleation in phase transitions, stochastic switching in gene regulatory circuits, or failure events in synthetic biochemical circuits.
In conclusion, the authors provide a clear derivation of how intrinsic reaction noise, captured by the chemical Langevin equation, can be harnessed within Kramers escape theory to predict the rate of rare, potentially catastrophic events. Their analytical treatment of the skin cell proliferation model not only validates the approach against previous numerical findings but also yields insight into how model parameters control the exponential suppression of uncontrolled growth. This work thus bridges stochastic chemical kinetics with classical escape theory, opening avenues for predictive modeling of rare events across chemistry, biology, and materials science.
Comments & Academic Discussion
Loading comments...
Leave a Comment