The electronic structure and dipole moment of charybdotoxin, a scorpion venom peptide with K+ channel blocking activity
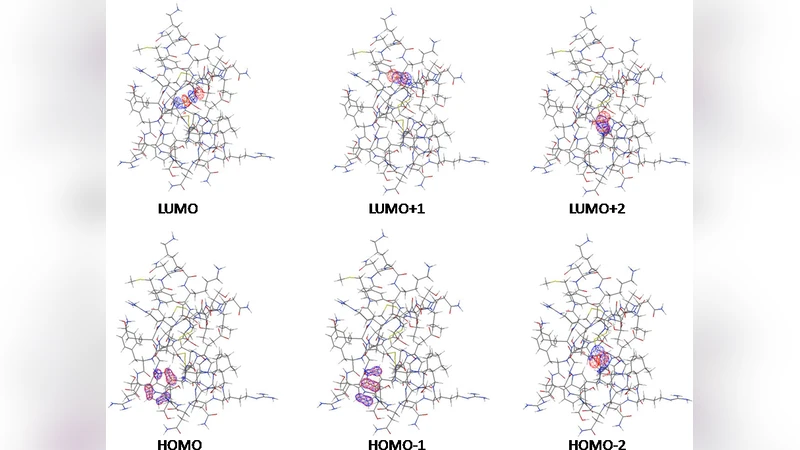
The electronic structure of charybdotoxin (ChTX), a scorpion venom peptide that is known to act as a potassium channel blocker, is investigated with the aid of quantum mechanical calculations. The dipole moment vector (145 D) of ChTX can be stirred by the full length KcsA potassium channel’s macrodipole (403 D) thereby assuming the proper orientation before binding the ion channel on the cell surface. The localization of the frontier orbitals of ChTX has been revealed for the first time. HOMO is localized on Trp14 while the three lowest-energy MOs (LUMO, LUMO+1, and LUMO+2) are localized on the three disulfide bonds that characterize this pepetide. An effective way to engineer the HOMO-LUMO (H-L) gap of ChTX is that of replacing its Trp14 residue with Ala14 whereas deletion of the LUMO-associated disulfide bond with the insertion of a pair of L-alpha-aminobutyric acid residues does not affect the H-L energy gap.
💡 Research Summary
The paper presents a quantum‑mechanical investigation of charybdotoxin (ChTX), a 37‑residue scorpion‑venom peptide that blocks potassium channels. Using density‑functional theory (DFT) calculations, the authors first optimized the full atomic structure of ChTX, including its three disulfide bridges, and then computed its electronic properties in both vacuum and a polarizable continuum solvent model. The most striking result is the magnitude of the peptide’s permanent dipole moment: 145 Debye (D), a value that is unusually large for a peptide of this size.
To place this dipole in a physiological context, the authors modeled the electrostatic field generated by the full‑length KcsA potassium channel, whose own macrodipole has been previously calculated as 403 D. By treating the channel’s dipole as a uniform electric field, they showed that the torque exerted on ChTX is sufficient to rotate the peptide so that its dipole vector aligns with the channel’s entrance. This “dipole‑steering” mechanism provides a plausible physical explanation for how ChTX can adopt the correct orientation on the cell surface before establishing the high‑affinity contacts that block ion conduction. It extends the classic notion of charge complementarity by demonstrating that long‑range electrostatic forces can pre‑orient a toxin even before specific side‑chain interactions occur.
The frontier molecular orbital (FMO) analysis revealed a clear spatial segregation of the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbitals (LUMO, LUMO+1, LUMO+2). The HOMO is localized on the indole side chain of Trp14, a residue that contributes a large π‑electron cloud and therefore dominates the electron‑donating character of the peptide. In contrast, the three lowest‑energy LUMOs are each centered on one of the three disulfide bonds (Cys‑Cys linkages). Disulfide bridges are known to be highly electron‑withdrawing; their involvement in the LUMO manifold explains why these bonds act as primary electron‑acceptor sites and may be crucial for the redox stability of the toxin.
Armed with this orbital map, the authors explored two rational design strategies aimed at modulating the HOMO‑LUMO energy gap (the H‑L gap), a key descriptor of chemical reactivity, optical absorption, and charge‑transfer propensity. The first strategy replaces Trp14 with alanine (Ala14). Removing the indole π system lowers the HOMO energy, thereby widening the H‑L gap by roughly 0.4 eV. This change is predicted to increase the peptide’s electronic stability and shift any absorption band toward shorter wavelengths, which could be advantageous for spectroscopic monitoring or for reducing off‑target photochemical reactions.
The second strategy targets the LUMO‑associated disulfide bonds. By deleting a specific S‑S bridge and inserting a pair of L‑α‑aminobutyric acid (α‑Abu) residues, the authors created a mutant in which the LUMO energy remains essentially unchanged. Consequently, the H‑L gap is preserved, while the overall backbone flexibility and possibly the proteolytic resistance of the peptide are altered. Importantly, the calculated dipole moment of this mutant differs by less than 10 % from that of the wild‑type toxin, indicating that the electrostatic steering effect would be retained.
The paper also compares the charge distribution and dipole orientation of the wild‑type and mutant toxins. The Trp→Ala substitution reduces the total dipole magnitude modestly but does not alter its direction, suggesting that the steering torque from the channel would still be effective. The disulfide‑to‑α‑Abu conversion leaves both dipole magnitude and direction virtually unchanged, reinforcing the idea that the macrodipole‑driven alignment is robust to certain structural modifications.
In summary, the study provides three major insights: (1) ChTX possesses a large intrinsic dipole that can be reoriented by the macrodipole field of a potassium channel, offering a physical basis for pre‑binding alignment; (2) the electronic frontier orbitals are spatially segregated, with the HOMO on Trp14 and the LUMOs on the disulfide bridges, highlighting distinct electron‑donor and electron‑acceptor regions within the toxin; and (3) targeted residue substitutions (Trp14→Ala) or disulfide‑bridge replacements (with α‑Abu) allow rational tuning of the HOMO‑LUMO gap without compromising the dipole‑driven steering mechanism. These findings not only deepen our mechanistic understanding of scorpion‑venom peptide inhibition of potassium channels but also open avenues for the rational engineering of peptide‑based channel blockers with tailored electronic, spectroscopic, and pharmacokinetic properties.
Comments & Academic Discussion
Loading comments...
Leave a Comment