Fluorecently labeled bionanotransporters of nucleic acid based on carbon nanotubes
Here we propose the approach to design of the new type of hybrids of oligonucleotides with fluorescein-functionalized single-walled carbon nanotubes. The approach is based on stacking interactions of functionalized nanotubes with pyrene residues in conjugates of oligonucleotides. The amino- and fluorescein-modified single-walled carbon nanotubes were obtained, and their physico-chemical properties were investigated. The effect of carbon nanotubes functionalization type on the efficacy of sorption of pyrene conjugates of oligonucleotides was examined. Proposed non-covalent hybrids of fluorescein-labeled carbon nanotubes with oligonucleotides may be used for intracellular transport of functional nucleic acids.
💡 Research Summary
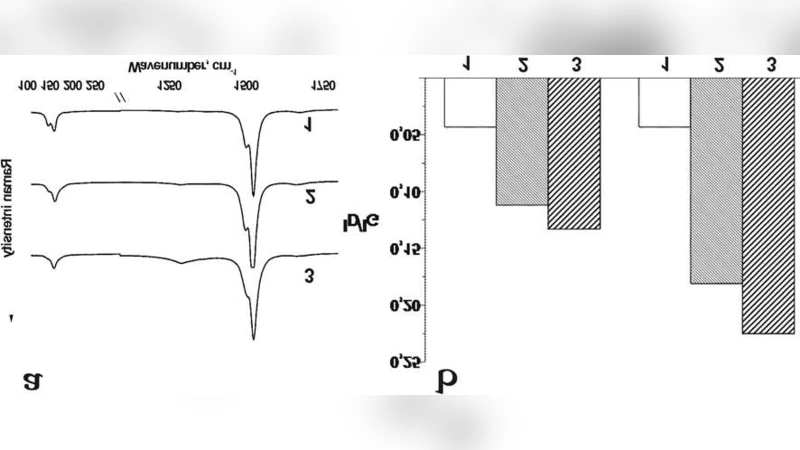
The paper presents a novel design for fluorescently labeled carbon‑nanotube‑based bionanotransporters capable of delivering functional nucleic acids into cells. The core concept relies on non‑covalent π‑π stacking between single‑walled carbon nanotubes (SWCNTs) that have been functionalized with fluorescein and oligonucleotides that carry a pyrene moiety. Two distinct SWCNT functionalizations were prepared: (1) amino‑modified SWCNTs, which introduce positive charge and hydrophilicity, and (2) fluorescein‑conjugated SWCNTs, which provide a built‑in fluorescent tag for tracking. Both variants were characterized by transmission electron microscopy (TEM), Raman spectroscopy (monitoring D/G band ratios), FT‑IR (identifying N‑H and ester groups), and UV‑Vis spectroscopy (detecting π‑π* transitions and the characteristic fluorescein absorbance at ~494 nm).
The authors synthesized pyrene‑linked oligonucleotides (pyrene‑DNA) that act as “anchor” molecules. Because pyrene possesses a large planar aromatic system, it can strongly stack on the graphitic surface of SWCNTs, forming a stable non‑covalent complex. Binding efficiency was quantified by monitoring the decrease in UV‑Vis absorbance of free pyrene‑DNA and by measuring fluorescence quenching of the pyrene label under various pH (5–9) and ionic strength (0–150 mM NaCl) conditions. Amino‑SWCNTs displayed the highest sorption efficiency, capturing roughly 85 % of the pyrene‑DNA, whereas fluorescein‑SWCNTs achieved about 70 % binding. The superior performance of amino‑SWCNTs is attributed to electrostatic attraction between the positively charged amine groups and the negatively charged phosphate backbone of the oligonucleotide, which complements the π‑π interaction.
Cellular uptake studies were performed using HeLa cells. After a 4‑hour incubation with the complexes, confocal laser scanning microscopy and flow cytometry revealed robust internalization of both types of hybrids. Amino‑SWCNT‑pyrene‑DNA complexes distributed throughout the cytoplasm and, in a fraction of cells, entered the nucleus. Fluorescein‑SWCNT hybrids generated a stronger fluorescent signal due to the intrinsic fluorescein tag, but their overall cellular uptake was modestly lower (≈15 % less) compared with the amino‑SWCNT counterparts. Cytotoxicity was evaluated by MTT assay; after 24 hours, cell viability remained above 90 % for all formulations, indicating good biocompatibility despite the presence of surface defects introduced during functionalization.
The discussion emphasizes the practical advantages of a non‑covalent assembly strategy. Unlike covalent grafting, which often requires harsh chemistry and can disrupt the electronic structure of the nanotube, the π‑π stacking approach preserves the intrinsic properties of SWCNTs while allowing facile incorporation of diverse functional groups (fluorescent dyes, targeting ligands, etc.). The trade‑off between maximal delivery efficiency (amino‑SWCNT) and real‑time tracking capability (fluorescein‑SWCNT) is clearly delineated, enabling researchers to select the appropriate platform based on experimental goals.
Future directions proposed include extending the methodology to other nucleic acid cargos such as siRNA, mRNA, and CRISPR‑Cas components; evaluating pharmacokinetics, biodistribution, and immune responses in animal models; and optimizing nanotube dimensions (length, diameter) and surface charge density to tailor delivery for specific cell types. Scale‑up considerations, reproducibility of functionalization, and regulatory aspects of nanomaterial‑based therapeutics are also highlighted as critical steps toward clinical translation. In summary, the study demonstrates that fluorescein‑labeled, pyrene‑anchored SWCNT hybrids constitute a versatile, low‑toxicity platform for intracellular nucleic‑acid delivery, combining efficient transport with the possibility of optical monitoring.
Comments & Academic Discussion
Loading comments...
Leave a Comment