An analysis of overall network architecture reveals an infinite-period bifurcation underlying oscillation arrest in the segmentation clock
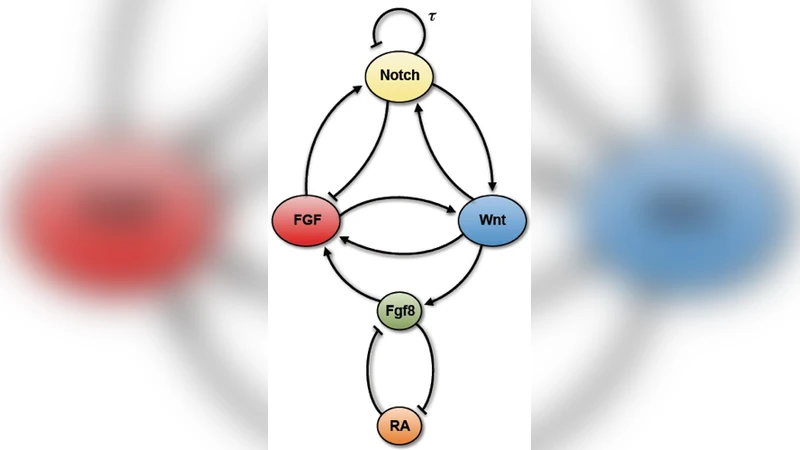
Unveiling the mechanisms through which the somitogenesis regulatory network exerts spatiotemporal control of the somitic patterning has required a combination of experimental and mathematical modeling strategies. Significant progress has been made for the zebrafish clockwork. However, due to its complexity, the clockwork of the amniote segmentation regulatory network has not been fully elucidated. Here, we address the question of how oscillations are arrested in the amniote segmentation clock. We do this by constructing a minimal model of the regulatory network, which privileges architectural information over molecular details. With a suitable choice of parameters, our model is able to reproduce the oscillatory behavior of the Wnt, Notch and FGF signaling pathways in presomitic mesoderm (PSM) cells. By introducing positional information via a single Wnt3a gradient, we show that oscillations are arrested following an infinite-period bifurcation. Notably: the oscillations increase their amplitude as cells approach the anterior PSM and remain in an upregulated state when arrested; the transition from the oscillatory regime to the upregulated state exhibits hysteresis; and an opposing distribution of the Fgf8 and RA gradients in the PSM arises naturally in our simulations. We hypothesize that the interaction between a limit cycle (originated by the Notch delayed-negative feedback loop) and a bistable switch (originated by the Wnt-Notch positive cross-regulation) is responsible for the observed segmentation patterning. Our results agree with previously unexplained experimental observations and suggest a simple plausible mechanism for spatiotemporal control of somitogenesis in amniotes.
💡 Research Summary
The paper addresses a long‑standing question in vertebrate development: how the oscillatory segmentation clock that drives somite formation in amniotes is arrested as cells move from the posterior to the anterior presomitic mesoderm (PSM). The authors adopt a “network‑architecture‑first” strategy, constructing a minimal mathematical model that captures the essential wiring of the mouse segmentation regulatory network while deliberately abstracting away most molecular details.
The model consists of five ordinary differential equations with a single explicit time delay (τ≈45 min) representing the delayed negative feedback in the Notch pathway, which is taken as the sole source of self‑sustained oscillations. The three signaling pathways—Wnt, Notch, and FGF—are represented by variables w, n, and f, respectively. Two additional variables, x and y, stand for the concentrations of Fgf8 and retinoic acid (RA). Interactions are encoded as additive Hill functions (cooperativity 4) with parameters β (maximal regulatory strength) and K (half‑saturation). Positive cross‑regulation between Wnt and Notch (β_wn, β_nw) is made dominant to generate a bistable switch, while Wnt‑FGF positive coupling and Notch‑FGF negative coupling are also included. Finally, the antagonistic double‑negative circuit between Fgf8 and RA is modeled with linear terms for the ligand‑receptor binding.
Positional information is introduced through a single scalar k (0 ≤ k ≤ 1) that mimics the decreasing Wnt3a gradient along the AP axis. The gradient influences the model in two ways: (i) it raises the degradation rate of the Notch intracellular domain (γ_n = 0.02 k + 0.13 min⁻¹), thereby weakening Notch signaling, and (ii) it reduces the production rate of Fgf8 (β_xw = 0.1 k min⁻¹), linking Wnt activity to the Fgf8/RA antagonism. As the embryo elongates, cells experience decreasing k values, moving from a high‑Wnt environment (k ≈ 1) to low‑Wnt (k ≈ 0).
Bifurcation analysis using k as the control parameter reveals a SNIC (saddle‑node on invariant circle) or infinite‑period bifurcation at k ≈ 0.44. Above this threshold the system exhibits a stable limit cycle with a period of ~120 min, matching mouse clock‑wave data. As k declines, the period lengthens and the oscillation amplitude grows—precisely the “slow‑down” and amplitude increase observed experimentally in anterior PSM cells. When k falls below the critical value, the limit cycle disappears and the system settles into a high‑Wnt/high‑Notch steady state with moderate FGF activity. Importantly, the reverse transition occurs at a higher k ≈ 0.76, producing a wide hysteresis loop that explains the irreversible nature of somite commitment.
The model also reproduces the opposing gradients of Fgf8 and RA. Because Wnt3a drives Fgf8 transcription, decreasing k suppresses Fgf8 while RA, which is negatively regulated by Fgf8, rises in the anterior PSM. This emergent pattern aligns with experimental observations and with earlier theoretical work on a bistable Fgf8/RA domain.
Overall, the study demonstrates that a minimal network comprising (1) a delayed negative feedback loop in Notch (oscillator) and (2) a positive Wnt‑Notch cross‑regulatory loop (bistable switch) is sufficient to generate the full repertoire of dynamical behaviors seen in amniote segmentation: sustained oscillations, gradual period lengthening, amplitude increase, hysteretic arrest, and the formation of antagonistic morphogen gradients. By focusing on architecture rather than detailed biochemistry, the authors provide a parsimonious yet biologically plausible explanation for how spatial cues (Wnt3a gradient) are translated into temporal control of the segmentation clock, offering a valuable framework for future experimental and theoretical investigations.
Comments & Academic Discussion
Loading comments...
Leave a Comment