Modelling an Ammonium Transporter with SCLS
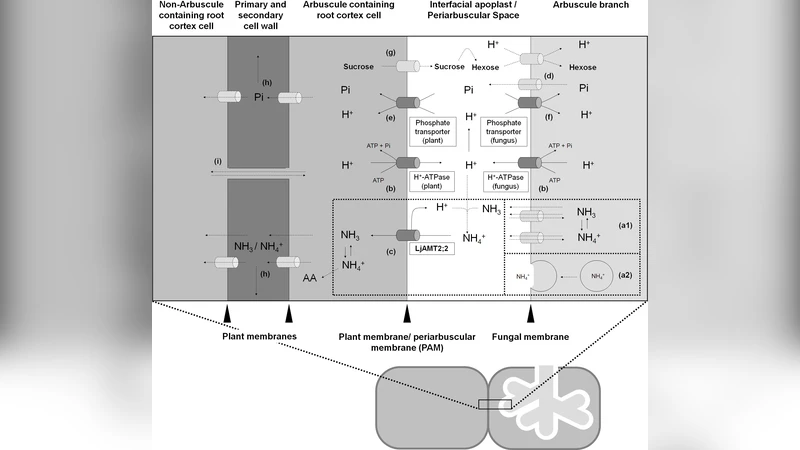
The Stochastic Calculus of Looping Sequences (SCLS) is a recently proposed modelling language for the representation and simulation of biological systems behaviour. It has been designed with the aim of combining the simplicity of notation of rewrite systems with the advantage of compositionality. It also allows a rather simple and accurate description of biological membranes and their interactions with the environment. In this work we apply SCLS to model a newly discovered ammonium transporter. This transporter is believed to play a fundamental role for plant mineral acquisition, which takes place in the arbuscular mycorrhiza, the most wide-spread plant-fungus symbiosis on earth. Due to its potential application in agriculture this kind of symbiosis is one of the main focuses of the BioBITs project. In our experiments the passage of NH3 / NH4+ from the fungus to the plant has been dissected in known and hypothetical mechanisms; with the model so far we have been able to simulate the behaviour of the system under different conditions. Our simulations confirmed some of the latest experimental results about the LjAMT2;2 transporter. The initial simulation results of the modelling of the symbiosis process are promising and indicate new directions for biological investigations.
💡 Research Summary
The paper presents a novel application of the Stochastic Calculus of Looping Sequences (SCLS) to model the transport of ammonium (NH₃/NH₄⁺) across the symbiotic interface between arbuscular mycorrhizal fungi and plant roots. The biological focus is the recently identified LjAMT2;2 transporter, which is thought to play a pivotal role in plant mineral acquisition by mediating the passage of nitrogenous compounds from the fungal partner to the host plant. The authors first decompose the functional cycle of LjAMT2;2 into three mechanistic steps—binding, translocation, and release—and encode each step as a stochastic rewrite rule within the SCLS formalism.
In SCLS, biological entities are represented as linear sequences and closed loops; the latter are used to model membranes. By treating the fungal cell, the plant cell, and the intervening membrane as separate but interacting subsystems, the authors preserve modularity while allowing material exchange through shared rewrite rules. The binding rule captures the association of external NH₃ with the transporter’s hydrophilic pocket (NH₃·T → T·NH₃). The translocation rule incorporates the effect of the membrane potential (−120 mV) by converting the bound species into a charged NH₄⁺ that moves across the loop (T·NH₄⁺ → NH₄⁺·T). The release rule is pH‑dependent, reflecting experimental observations that intracellular pH (≈7.2) influences whether NH₃ or NH₄⁺ is expelled into the plant cytosol.
Parameter values for membrane potential, extracellular and intracellular pH, and the NH₃/NH₄⁺ ratio were taken from recent experimental measurements. The stochastic simulation algorithm (SSA) based on Gillespie’s method was run for one million reaction events, providing time‑resolved concentration profiles for both NH₃ and NH₄⁺. The simulation outcomes reveal three key insights: (1) a stronger negative membrane potential favors the charged NH₄⁺ translocation pathway, confirming an electro‑driven component of LjAMT2;2 activity; (2) lower intracellular pH suppresses NH₃ release by promoting protonation to NH₄⁺, highlighting the transporter’s pH sensitivity; and (3) a stepwise translocation mechanism fits the experimental data better than a hypothesized simultaneous NH₃/NH₄⁺ flux, thereby supporting the authors’ mechanistic proposal.
Beyond the isolated transporter, the authors extend the model to the full symbiotic system. They construct a cascade of SCLS rules that describe NH₃ production by the fungal hyphae, diffusion through the apoplastic space, and subsequent uptake by LjAMT2;2 on the plant root membrane. The integrated model reproduces the characteristic “burst” of fungal NH₃ synthesis during early colonization, followed by a gradual increase in plant intracellular NH₄⁺ as the transporter becomes active. This behavior aligns with observed carbon‑nitrogen exchange dynamics in arbuscular mycorrhizae, suggesting that the SCLS framework can capture emergent properties of the whole symbiosis.
The authors conclude that SCLS offers a compact yet expressive language for modeling membrane‑bound transport processes, combining the simplicity of rewrite systems with the compositional power needed for multi‑compartment biological networks. Their LjAMT2;2 model not only validates recent experimental findings but also generates testable predictions about the influence of membrane potential and pH on ammonium flux. Future work will incorporate additional metabolic pathways, signaling cascades, and environmental variables such as soil nitrogen availability and temperature, aiming to provide a systems‑level platform for hypothesis generation and experimental design in plant‑fungus symbiosis research. The study demonstrates that formal computational models like SCLS can accelerate the translation of molecular insights into agronomic applications, particularly in the context of the BioBITs project’s goal to enhance sustainable nutrient acquisition in crops.
Comments & Academic Discussion
Loading comments...
Leave a Comment