Revealing latent factors of temporal networks for mesoscale intervention in epidemic spread
The customary perspective to reason about epidemic mitigation in temporal networks hinges on the identification of nodes with specific features or network roles. The ensuing individual-based control strategies, however, are difficult to carry out in practice and ignore important correlations between topological and temporal patterns. Here we adopt a mesoscopic perspective and present a principled framework to identify collective features at multiple scales and rank their importance for epidemic spread. We use tensor decomposition techniques to build an additive representation of a temporal network in terms of mesostructures, such as cohesive clusters and temporally-localized mixing patterns. This representation allows to determine the impact of individual mesostructures on epidemic spread and to assess the effect of targeted interventions that remove chosen structures. We illustrate this approach using high-resolution social network data on face-to-face interactions in a school and show that our method affords the design of effective mesoscale interventions.
💡 Research Summary
The paper tackles the challenge of designing epidemic mitigation strategies that go beyond individual‑based interventions, which are often impractical and ignore the intertwined temporal and topological patterns present in real contact networks. The authors propose a mesoscopic framework that identifies collective structures—referred to as “mesostructures”—at multiple scales and quantifies their influence on disease spread.
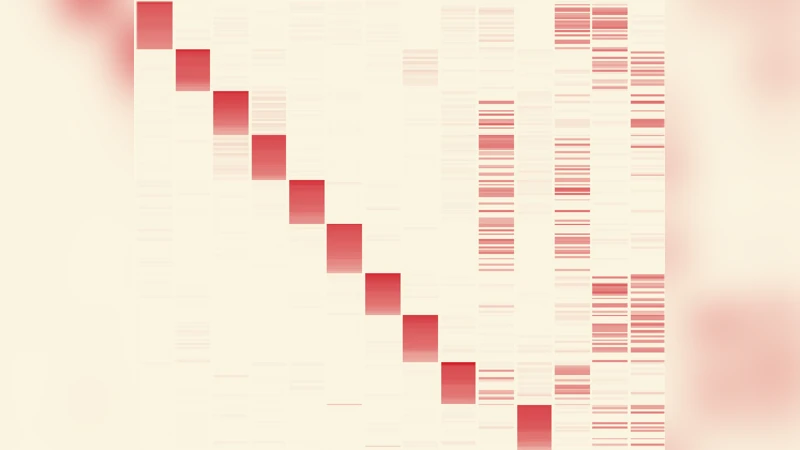
Methodologically, the authors represent a temporal network as a three‑dimensional tensor T(N × N × L), where N is the number of nodes and L the number of discrete time slices. Using non‑negative tensor factorization (NTF), they approximate T as a sum of R rank‑1 tensors S_r = a_r ⊗ a_r ⊗ c_r. The vector a_r captures the participation weight of each node in mesostructure r, while c_r describes its temporal activation profile. The non‑negativity constraint yields a parts‑based decomposition that is readily interpretable.
The authors apply this pipeline to a high‑resolution dataset of face‑to‑face contacts collected in an Italian primary school. The data comprise 241 individuals (students and teachers) observed over two consecutive days, aggregated into 15‑minute snapshots, resulting in a tensor with N = 241 and L = 70. NTF with R = 14 components reveals two families of mesostructures: (i) ten components (S₁‑S₉, S₁₁) that correspond to the ten school classes—stable, cohesive groups that can also be detected by conventional community‑detection on static aggregates; and (ii) four components (S₁₀, S₁₂‑S₁₄) that mix members of different classes but are active only during lunch breaks, representing temporally localized mixing events that static methods miss. Each component’s contribution to the total tensor weight is reported, providing a baseline for comparison.
To assess the functional relevance of each mesostructure, the authors construct altered networks \tilde T^{(−r)} by removing a single component S_r from the sum and recombining the remaining terms. They then simulate two epidemic processes on both the original network \tilde T and each altered network: (a) a simple Susceptible‑Infected (SI) model, where infection spreads with probability λ per contact and never recovers; and (b) a more realistic Susceptible‑Infected‑Recovered (SIR) model with infection rate λ and recovery rate μ. For the SI model they compute a “delay ratio” τ_r = ⟨t_r − t⟩/⟨t⟩, where t is the half‑infection time on the full network and t_r on the altered network. For the SIR model they compute a size ratio ρ_r = ⟨Ω_r⟩/⟨Ω⟩, where Ω and Ω_r are the final numbers of infected or recovered individuals.
The results reveal that not all components are equally important for spreading. Removing the class‑based components (which carry a large fraction of the total weight) yields modest increases in τ_r and modest reductions in ρ_r, comparable to what is observed when the same amount of weight is removed at random. In stark contrast, eliminating the lunch‑time mixing components—despite their relatively small weight—produces a pronounced slowdown of the SI process (large τ_r) and a substantial reduction of the final epidemic size in the SIR simulations (low ρ_r). This demonstrates that temporally concentrated cross‑class interactions act as critical bridges for disease propagation.
To ensure that the observed effects are not merely due to a reduction in overall contact volume, the authors generate null models by randomly deleting a set of edges whose total weight matches that of the removed component. The empirical τ_r and ρ_r values for the lunch‑time components lie far outside the null distributions, confirming that their impact stems from specific structural‑temporal patterns rather than from a simple loss of contacts.
The study thus provides a concrete methodological pipeline: (1) encode a temporal contact network as a tensor; (2) apply NTF to extract interpretable mesostructures; (3) reconstruct counterfactual networks by removing selected components; (4) evaluate epidemic outcomes on original versus altered networks; and (5) rank mesostructures by their dynamical importance. The authors argue that such mesoscopic interventions—e.g., restricting or reshaping activities during identified high‑risk periods like lunch breaks—can achieve substantial epidemic mitigation while avoiding the socio‑economic costs of blanket measures such as full school closures.
In conclusion, the paper demonstrates that tensor‑based latent factor analysis can uncover hidden, time‑localized mixing patterns that dominate disease spread, and that targeted, intermediate‑scale interventions informed by this analysis are both effective and operationally feasible. The framework is generalizable to other settings (workplaces, public transport, etc.) and opens avenues for real‑time monitoring and adaptive control of epidemics in temporally evolving contact networks.
Comments & Academic Discussion
Loading comments...
Leave a Comment