The enzyme cost of given metabolic flux distributions, as a function of logarithmic metabolite levels, is convex
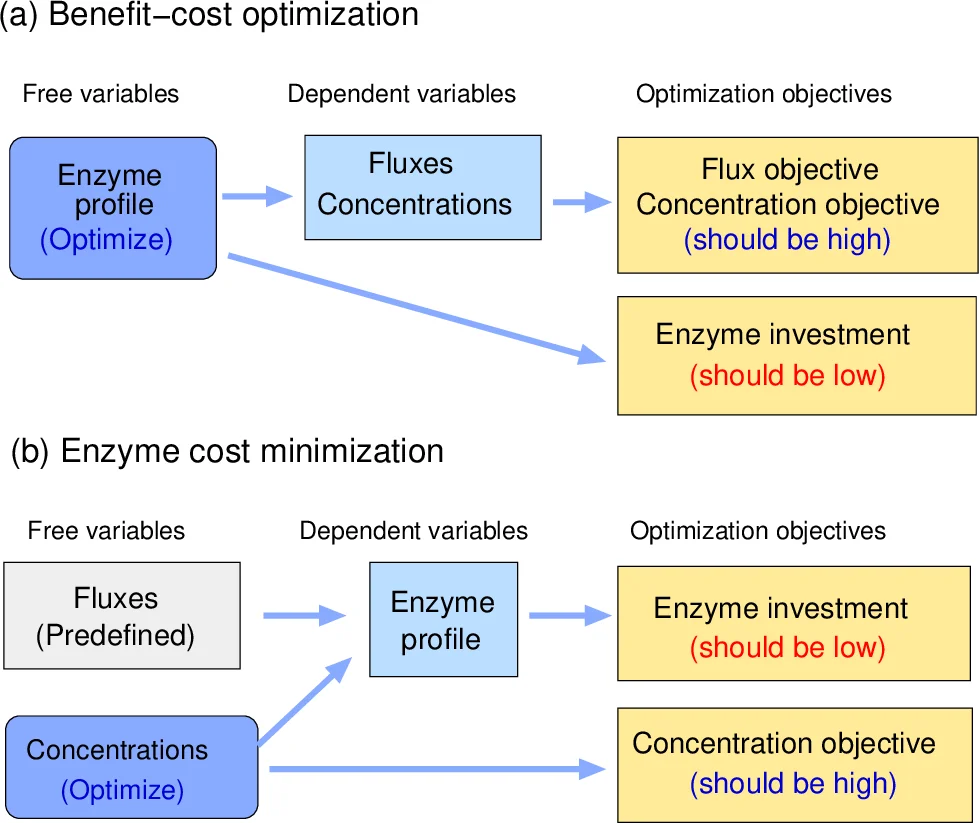
Enzyme costs play a major role in the choice of metabolic routes, both in evolution and bioengineering. Given desired fluxes, necessary enzyme levels can be estimated based on known rate laws and on a principle of minimal enzyme cost. With logarithmic metabolite levels as free variables, enzyme cost functions and constraints in optimality and sampling problems can be handled easily. The set of feasible metabolite profiles forms a polytope in log-concentration space, whose points represent all possible steady states of a kinetic model. We show that enzyme cost is a convex function on this polytope. This makes enzyme cost minimization - finding optimal enzyme profiles and corresponding metabolite profiles that realize a desired flux at a minimal cost - a convex optimization problem.
💡 Research Summary
The paper addresses the problem of determining the enzyme levels required to sustain a prescribed set of metabolic fluxes, taking into account realistic kinetic rate laws and a principle of minimal enzyme cost. By treating the logarithms of metabolite concentrations as free variables, the authors show that the feasible set of metabolite profiles forms a convex polytope in log‑concentration space. Within this polytope, each point corresponds to a possible steady state of the kinetic model, linking metabolite levels to the enzyme amounts needed to achieve the target fluxes.
A central contribution is the factorization of reversible rate laws into a product of four terms: the forward catalytic constant (k_cat), a thermodynamic efficiency (η_th = 1 − e^{−Θ}), a kinetic efficiency (η_kin, derived from the denominator of the rate law), and an optional regulatory efficiency (η_reg). This “separable rate law” allows the enzyme demand for reaction l to be expressed as ε_l = v_l / (k_cat,l · η_th,l · η_kin,l · η_reg,l). Multiplying by a reaction‑specific cost coefficient h_l yields the enzyme cost y_l, and the total cost for a pathway is y_pw = ∑_l y_l.
The authors then define the metabolite polytope P by two types of linear constraints: (i) upper and lower bounds on each log‑concentration (forming a box) and (ii) thermodynamic driving force constraints Θ_l · v_l > 0, which translate into linear inequalities in log‑space. Faces where Θ_l = 0 (E‑faces) correspond to equilibrium and cause the cost to diverge, while the physiological bounds (P‑faces) truncate the polytope.
The key mathematical result is that the enzyme cost function y_pw(x) is convex on P. This follows because η_th^{-1} is convex in the linear combination Θ (which itself is linear in x), and η_kin^{-1} is convex because it is the reciprocal of a positive polynomial in the exponentials of x. Consequently, each term h_l·v_l/(k_cat,l·η_th,l·η_kin,l) is a convex function of x, and their sum preserves convexity. Even if the cost is not strictly convex (e.g., flat regions), adding a convex regularization term such as y_reg = ∑ γ_i (x_i − x_i^0)^2 restores strict convexity, guaranteeing a unique global optimum.
With convexity established, the enzyme cost minimization (ECM) problem becomes a standard convex optimization:
y_opt(v) = min_{x∈P} y_pw(x),
x_opt(v) = argmin_{x∈P} y_pw(x),
ε_opt(v) = ε(v, c_opt(v)).
These can be solved efficiently using interior‑point methods, gradient‑based algorithms, or any convex solver. The optimal solution provides both the metabolite concentrations (in log‑space) and the corresponding minimal enzyme levels required to sustain the desired flux distribution.
The paper discusses practical implications: unlike Flux Balance Analysis (FBA), which ignores enzyme kinetics and costs, the ECM framework incorporates thermodynamic feasibility, substrate saturation, and enzyme-specific costs, yielding more realistic predictions for metabolic engineering and evolutionary studies. It also allows extensions to multi‑objective optimization, for example by adding a concave metabolite‑level objective z_met(x) to favor concentrations near physiological “ideal” values.
In summary, the authors demonstrate that when metabolite concentrations are expressed logarithmically, the enzyme cost associated with any feasible flux distribution is a convex function over a convex polytope of metabolite states. This insight converts the otherwise complex problem of enzyme allocation into a tractable convex optimization, opening the door to systematic, computationally efficient design of metabolic pathways with minimal enzyme investment.
Comments & Academic Discussion
Loading comments...
Leave a Comment