Static Analysis for Biological Systems (BioAmbients)
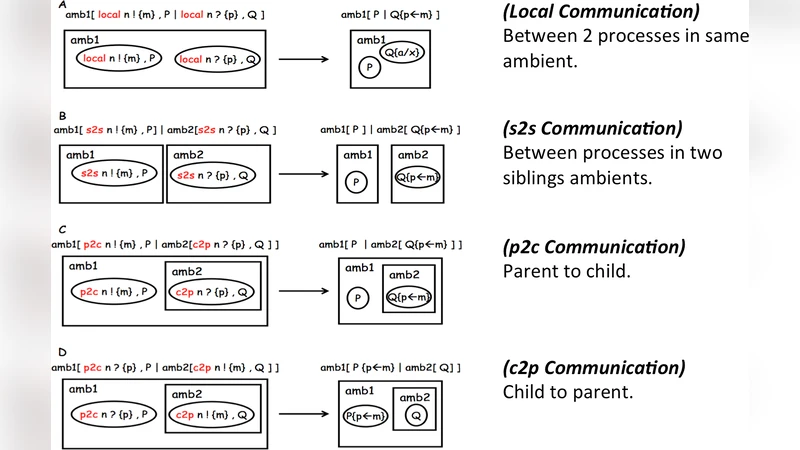
In this paper, I present a summary on some works that utilized static analysis techniques for understanding biological systems. Control flow analysis, context dependent analysis, and other techniques were employed to investigate the properties of BioAmbients. In this summary report, I tried to introduce the ideas and explain the techniques used in the subject papers. This summary will highlight the biological concepts of BioAmbients.
💡 Research Summary
The paper provides a comprehensive review of how static analysis techniques have been applied to the BioAmbients formalism for modeling biological systems. BioAmbients extend the Ambient Calculus to capture mobile compartments such as organelles, protein complexes, and signaling domains, representing their movement, composition, and communication in a mathematically rigorous way. Because biological networks are often too large and complex for exhaustive simulation, static analysis offers a pre‑execution method to over‑approximate possible behaviors, prune infeasible interactions, and guarantee safety properties.
Three principal static‑analysis approaches are examined. First, Control‑Flow Analysis (CFA) constructs a name‑space and channel mapping for each ambient, then propagates movement and communication rules to generate a graph of all potential inter‑ambient communications. This over‑approximation enables early detection of impossible communication paths and, in case studies, predicts that aberrant protein transport can trigger apoptosis pathways before any wet‑lab experiment.
Second, Context‑Dependent Analysis (CDA) introduces environment labels that annotate each ambient with the cellular region it occupies (e.g., cytosol, lysosome). Label‑propagation rules capture how a change in context alters the ambient’s internal actions. CDA is shown to be valuable for drug‑delivery design, allowing researchers to verify that a nanocarrier will only release its payload in the target tissue while remaining inert elsewhere.
Third, Hybrid Static‑Dynamic Analysis combines the conservative over‑approximation of static methods with empirical data from simulations or experiments. Static invariants are paired with probabilistic transition matrices derived from dynamic runs, yielding quantitative estimates of the likelihood that a system reaches an error state. This hybrid approach was applied to a large metabolic network to identify bottlenecks and to prioritize cost‑effective experimental validations.
The paper emphasizes that static analysis not only aids in model verification and error detection but also guides hypothesis generation. By revealing unreachable states, context‑specific behaviors, and probabilistic risk assessments, researchers can focus experimental resources on the most informative scenarios.
Future directions highlighted include the integration of richer type systems (e.g., probabilistic or dependent types), Bayesian inference mechanisms, and machine‑learning‑based approximations to improve precision while retaining scalability. Moreover, establishing a feedback loop where experimental results continuously refine static analyses could lead to self‑optimizing models that evolve alongside biological discovery. In sum, the surveyed static‑analysis techniques demonstrate that BioAmbients can become a practical, mathematically grounded tool for systems biology, synthetic biology, and drug development.