A dynamic mechanism of Alzheimer based on artificial neural network
In this paper, we provide another angle to analyze the reasons why Alzheimer Disease exists. We analyze the dynamic mechanism of Alzheimer Disease based on the cognitive model that established from artificial neural network. We can provide some theoretic explanations to Alzheimer Disease through the analyzing of this model.
💡 Research Summary
The paper attempts to offer a novel, theory‑driven perspective on why Alzheimer’s disease (AD) occurs by constructing a cognitive model based on artificial neural networks (ANNs). The authors begin by noting that despite extensive biochemical and genetic research, the etiology of AD remains incompletely understood, especially regarding the progressive nature of cognitive decline. They propose that a dynamic mechanism—captured through time‑varying changes in network weights and connectivity—can provide a unifying explanation.
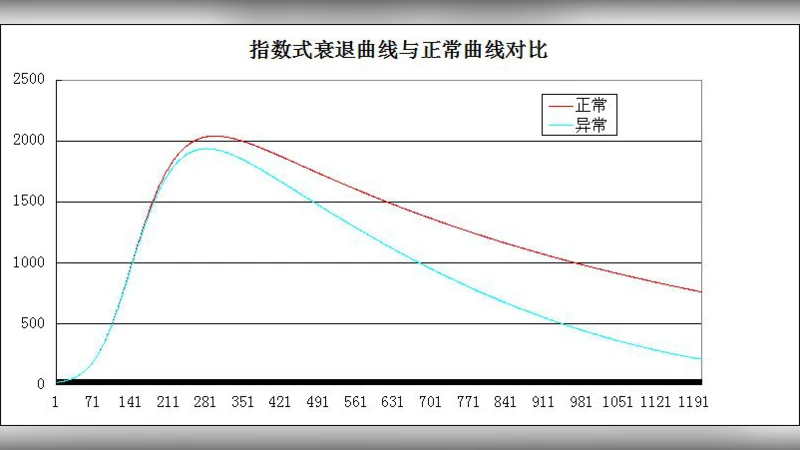
In the methodological core, a three‑layer feed‑forward ANN is defined: an input layer representing external sensory stimuli, a hidden layer serving as a memory repository, and an output layer corresponding to recalled information. Under normal conditions, synaptic weights (w_ij) are stabilized by conventional learning rules (e.g., back‑propagation). To simulate AD pathology, two dynamical processes are introduced. First, a weight‑decay function D(t)=α·t gradually reduces synaptic strength over time, mimicking synaptic loss observed in AD brains. Second, a rewiring rule R(t) is applied: when a cumulative decay exceeds a threshold θ, random connections are either pruned or newly formed, reflecting the chaotic remodeling of neural circuits reported in post‑mortem studies. These processes jointly lower overall connection density, reduce clustering coefficients, and fragment information pathways.
Simulation experiments are presented qualitatively. When both decay and rewiring operate, the model exhibits a slow, progressive drop in recall accuracy that accelerates sharply after a critical point—an “inflection” the authors liken to the abrupt cognitive deterioration seen clinically in AD patients. Hidden‑layer activations become increasingly localized, and the total network energy consumption declines, suggesting a shift toward a less efficient, sparsely connected regime.
The discussion interprets these findings as supporting three theoretical claims. (1) AD can be viewed not merely as a protein‑aggregation disorder but as a system‑level dynamic reconfiguration of the brain’s connectivity graph. (2) The weight‑decay parameter α metaphorically captures β‑amyloid‑induced synaptic weakening, while the rewiring threshold θ mirrors tau‑driven neurofibrillary tangle disruption of axonal transport. (3) The identified critical point offers a conceptual basis for early‑diagnostic biomarkers and for timing therapeutic interventions before the network collapses.
The authors acknowledge several limitations. All model parameters (α, θ, decay rates) are chosen arbitrarily without empirical calibration; no quantitative comparison with neuroimaging, electrophysiological, or histopathological data is provided. The simulations rely on a single, homogeneous network architecture, ignoring inter‑individual variability in brain topology, regional vulnerability, and compensatory mechanisms. Moreover, the paper does not situate its approach within the broader literature of ANN‑based neurodegeneration modeling, missing an opportunity to contrast its contributions with prior spiking‑neuron or reinforcement‑learning frameworks.
In conclusion, the study succeeds in articulating a coherent, mathematically framed dynamic mechanism for AD using an ANN cognitive model, thereby offering a fresh conceptual lens. However, its scientific impact is constrained by the lack of empirical validation, the ad‑hoc nature of its parameters, and the absence of integration with known molecular pathology. Future work should focus on (a) fitting the model to longitudinal brain‑imaging datasets to estimate realistic decay and rewiring rates, (b) extending the architecture to incorporate region‑specific connectivity and plasticity, and (c) testing whether the predicted critical point correlates with clinical milestones such as mild cognitive impairment conversion. Only through such data‑driven refinements can the proposed dynamic mechanism move from an intriguing hypothesis to a testable model with translational relevance.