Thermodynamics of statistical inference by cells
The deep connection between thermodynamics, computation, and information is now well established both theoretically and experimentally. Here, we extend these ideas to show that thermodynamics also places fundamental constraints on statistical estimat…
Authors: Alex H. Lang, Charles K. Fisher, Thierry Mora
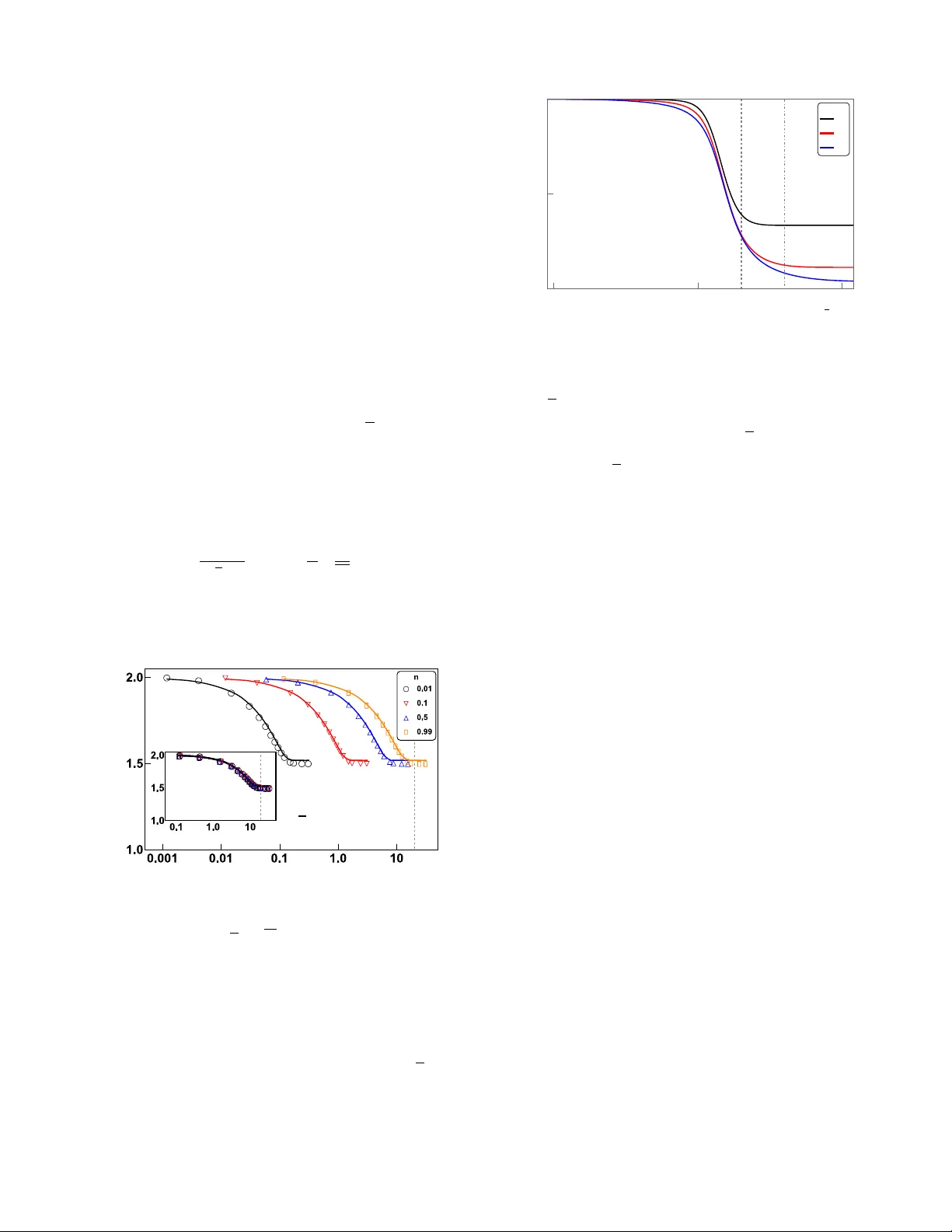
Thermo dynamics of statistical inference b y cells Alex H. Lang, 1 Charles K. Fisher, 1 Thierry Mora, 2 and Pank a j Mehta 1 1 Physics Dep artment, Boston University, Boston, Massachusetts 02215, USA 2 L ab or atoir e de physique statistique, CNRS, UPMC and ´ Ec ole normale sup´ erieur e, 75005 Paris, F r anc e The deep connection b et w een thermo dynamics, computation, and information is no w well estab- lished b oth theoretically and exp erimentally . Here, we extend these ideas to show that thermo- dynamics also places fundamental constraints on statistical estimation and learning. T o do so, w e in vestigate the constrain ts placed b y (nonequilibrium) thermodynamics on the abilit y of biochemical signaling netw orks to estimate the concen tration of an external signal. W e sho w that accuracy is limited by energy consumption, suggesting that there are fundamen tal thermo dynamic constraints on statistical inference. Cells often p erform complex computations in resp onse to external signals. These computations are implemen ted using elab orate bio c hemical netw orks that may op erate out of equilibrium and consume energy [1–7]. Given that energetic costs place imp ortant constrain ts on the design of ph ysical computing devices [8] and neural comput- ing architectures [9], one may conjecture that thermo- dynamic constraints also influence the design of cellular information pro cessing netw orks. This raises in tere sting questions ab out the relationship b etw een the informa- tion pro cessing capabilities of biochemical net works and energy consumption [10–14]. Indeed, we will sho w that thermo dynamics places fundamental constraints on the abilit y of bio chemical net works to perform statistical in- ference. More generally , statistical inference is intimately tied to the manipulation of information and hence offers a rich setting to study the relationship b et ween informa- tion and thermo dynamics [15–19]. In order for a cell to formulate an appropriate re- sp onse to an environmen tal signal, it m ust first estimate the concentration of an external signaling molecule us- ing membrane b ound receptors [1–6, 20]. The biophysics and biochemistry of cellular receptors is highly v ariable. Whereas some simple receptor proteins behav e lik e tw o- state systems (i.e. unbound and ligand bound) with dy- namics ob eying detailed balance [21], other receptors, suc h as G-protein coupled receptors (GPCRs), can ac- tiv ely consume energy as they cycle through multiple states. This naturally raises questions ab out ho w en- ergy consumption b y cellular receptors affects their abil- it y to perform statistical inference. Here, w e address these questions by analyzing the accuracy of statistical inference (i.e. learning) as a function of energy consump- tion in a simple but biophysically realistic mo del. W e sho w that learning more accurately alwa ys requires ex- p ending more energy , suggesting that the accuracy of a statistical estimator is fundamentally constrained by thermo dynamics. Cells estimate the concen tration of an external ligand using ligand-sp ecific receptors expressed on the cell sur- face. A ligand (usually a small molecule), at a con- cen tration c in the environmen t, binds the receptor at a concentration-dependent rate, k + c , and unbinds at a concen tration-indep enden t rate, k − [1] (see Fig. 1A). Up on ligand binding, the receptor protein undergo es con- formational changes or chemical mo difications that alter its activity , sending a signal that the ligand is b ound to do wnstream p ortions of the bio c hemical netw ork. Dur- ing a time in terv al T , the receptor can undergo multiple sto c hastic transitions b etw een the unbound nonsignaling state and the b ound signaling states. This information is con tained in the time series of signaling and nonsignaling in terv als (see Fig. 1B). After a time T , the cell con v erts this time series into an estimate for the external con- cen tration. A longer time series T alw ays giv es a b etter estimate for the concen tration; how ev er the cell needs to mak e a decision in a finite time, so w e consider T to be E. F. G. ADP ADP AT P AT P signal signal signal 0 0 1 2 1 k 10 k 10 k 01 k 01 k 21 k 12 k 02 k 20 signal signal signal 0 1 2 L k 10 k 01 k 21 k 12 k 0L k L0 A. k + c k - time Signaling Non- signaling T B. C. D. 𝞃 S 𝞃 NS FIG. 1: Schematic of a cell receptor and our mo del of a re- ceptor. (A) A c hemical ligand at concentration c binds to the receptor at rate k + c and unbinds at rate k − . (B) Ex- ample time series of a receptor binding. While unbound, the receptor is in nonsignaling state, but upon ligand binding it transitions to a signaling state. After a long time T , the recep- tor has a series of nonsignaling times τ N S and signaling times τ S from whic h to estimate the concen tration. (C) Two-state and (D) three-state bio c hemical models of a receptor. Up on ligand binding the receptor undergo es a ph ysical change (rep- resen ted as a conformational c hange) that transmits signals to the downstream bio c hemical net work. (E) Tw o-state, (F) three-state, and (G) L -state Mark ov mo dels of a receptor, where the c hain of states 3, 4, . . . L − 1 has b een suppressed. 2 fixed to a large but finite v alue. In principle, the esti- mate for the concen tration could b e computed using one of many different statistics that can b e obtained from this time series (e.g. a verage b ound time, av erage un- b ound time, etc.). Each of the resulting estimators for the external ligand concentration has a different accu- racy . F ollo wing Berg and Purcell (BP) [1], we measure the accuracy of an estimator for the concen tration using its “uncertain ty ,” defined as: uncertain ty := h ( δ c ) 2 i c 2 (1) where c is the mean and h ( δ c ) 2 i is the v ariance of the estimated concen tration. Sev eral methods ha ve b een proposed for how a cell ma y estimate the concentration of the external signaling molecule. In their pioneering pap er, Berg and Purcell suggested estimating the concen tration using the a verage time the receptor w as bound during the time T [1]. They sho wed that the minimal uncertain ty a receptor could ac hieve with this estimator was h ( δ c ) 2 i c 2 = 2 N (2) where N is the exp ected num b er of binding ev ents during the time interv al T . F or 30 y ears, man y thought that the BP estimator placed a fundamen tal limit on the accu- racy of a cellular receptor. How ever, in 2009, Endres and Wingreen [3] show ed that a cell using maxim um likeli- ho od estimation (MLE) based on the av erage nonsignal- ing time could reduce its uncertaint y b y half to h ( δ c ) 2 i c 2 = 1 N . (3) Ho wev er, the increased accuracy of MLE comes at an en- ergetic cost. Previous w ork [5] established that BP sets a limit for the b est p ossible estimator in equilibrium, im- plying that any receptor that performs MLE must op er- ate out of equilibrium and consume energy . In order to study the relationship b et ween thermody- namics and the accuracy of statistical estimators, we in- tro duce a new family of biophysically inspired cellular receptors that interpolate b et ween BP and MLE. In our mo del, receptors can activ ely consume energy by op erat- ing out of equilibrium (for example by hydrolyzing adeno- sine triphosphate or A TP). Using this family of models, w e show that there is a direct connection b et w een the energy consumed by a receptor and the uncertaint y of the resulting estimator. W e find that in order to learn more information (decrease its uncertaint y), the receptor m ust alwa ys exp end more energy (increase en tropy pro- duction). Note that, in this pap er, we restrict ourselves to mo deling the receptor and ignore the downstream sig- naling net w ork that con verts the signal from the receptor in to a cellular resp onse [10, 13]. Th us, the energies com- puted here represent low er b ounds on the total energy consumed b y the statistical estimation netw ork. Fig. 1C sho ws the simple tw o-state receptor considered b y BP . The binding of an external ligand to the receptor induces a change in the receptor from a nonsignaling state to a signaling state (see Fig 1B). The dynamics of this simple t wo-state receptor alwa ys obey detailed balance. Th us, in order to m odel nonequilibrium receptors, we m ust consider receptors with more than t w o states. Fig 1D sho ws a receptor with three states: one nonsignaling state to which ligands can bind, and t wo signaling states to which ligands cannot bind. With this extra state, the dynamics of the receptor can break detailed balance b y coupling the conformational change in the receptor to another reaction such as the hydrolosis of A TP . In particular, by consuming energy it is p ossible to drive the system preferentially through a series of state changes [22], for example clo c kwise in Fig 1F and Fig 1G. This results in a nonzero probabilit y flux through the state space and p ositive entrop y pro duction. In order to relate the thermodynamic properties of these receptors to their abilit y to p erform statistical in- ferences, it is useful to represent receptors as Marko v c hains. F or example, the tw o-state receptor shown in Fig. 1C can b e represented as a t wo-state Mark ov c hain with a state 0 corresp onding to the un bound nonsignal- ing state and state 1 corresp onding to the signaling state (see Fig. 1E). W e choose the transition rates b etw een states in the Mark ov chain to b e identical to the tran- sition rates b etw een conformations of the receptor. The three-state receptor can also b e mo deled as a three-state Mark ov chain with a ring structure, with state 0 once again corresp onding to the unbound, nonsignaling state (Fig. 1F). In this more abstract notation, it is easy to generalize the three-state receptor considered abov e to a receptor with L + 1 states (see Fig. 1G): L of these states are signaling states that cannot bind the ligand, while the remaining state, 0, corresp onds to the nonsignaling state that can bind ligands. F or ease of analysis, in this pap er, w e consider receptors arranged in a ring only . How ever, our mo del is a go od approximation for more complicated receptors with m ultiple path wa ys, so long as the receptor has a single path (for example, of length L ∗ ) that dom- inates the probability flux, see [23] for details. In that case, the complicated receptor reduces to a single ring of length L ∗ . A straightforw ard calculation shows that for the arc hi- tectures in Fig 1 [24], the uncertain ty of an estimate for the concen tration is given by [3]: h ( δ c ) 2 i c 2 = 1 N 1 + h ( δ τ S ) 2 i τ 2 S ≡ E N (4) where N is the num b er of binding even ts, τ S is the mean time spent in the signaling state after binding a ligand, 3 and h ( δτ S ) 2 i is the v ariance of the time sp ent in the sig- naling states. In the second equality , we ha ve defined the co efficien t E which measures the accuracy of an estima- tor; e.g. E = 2 for the Berg-Purcell limit and E = 1 for MLE. F or a given estimator (i.e. a specific arc hitecture and a set of rates ~ k ), we can calculate the mean and the v ariance of the signaling time by a first passage calcula- tion similar to that in [24, 25]. Here w e pro vide some in tuition for Eq. (4). Notice that all the information about the ligand concentration is contained in the even t of a ligand binding to the re- ceptor, and the un binding of the ligand, or the exiting of the signaling state, is indep endent of concentration. Th us, any v ariation in the duration of the signaling state adds additional noise to the estimate but do es not contain an y more information ab out the concentration. There- fore, the optimal estimator is one where the signaling in terv als are completely deterministic and h ( δ τ S ) 2 i = 0. Comparing Eqs. (4) and (3), we see that this corresponds to MLE. This is consistent with the well-kno wn fact that MLE is the optimal unbiased estimator for large sam- ple sizes. When the durations of the signaling times are exp onen tially distributed, like for a tw o-state receptor, h ( δ τ S ) 2 i = τ 2 S , then Eq. (4) reduces to the BP re- sult given in Eq. (2). Finally , in all cases, the uncer- tain ty scales in versely with the a verage n umber of bind- ing ev en ts N during the time interv al T . This scaling la w follo ws from the central limit theorem by treating each binding even t as an indep endent sample of the concen- tration. The Mark o v represen tation allo ws us to calculate the energy consumption using ideas from nonequilibrium sta- tistical ph ysics. W e focus on long time interv als, T 1, with man y binding ev ents, where the receptor dynamics can be mo deled b y nonequilibrium steady states (NESS). The en tropy pro duction of the Mark o v pro cess is the energy p er unit time (p o wer) required to main tain this NESS, and therefore calculating the en trop y pro duction is equiv alen t to calculating the energy consumed by the bio c hemical net work[10, 22]. The en trop y production is giv en b y [26] e p = L X i =0 L X j 6 = i p ss i k ij ln k ij k j i , (5) with p ss i is the steady state probability of state i , k ij is the transition rate from state i to state j , and we hav e set k B T = 1 [24]. F or the architectures where the Marko v pro cess forms a ring, the entrop y pro duction simplifies to e p = ( p ss 0 k 01 − p ss 1 k 10 ) ln k 01 k 12 ...k L 0 k 0 L k 10 ...k L,L − 1 = J ln γ (6) where J is the net flux around the ring and ln γ is the free energy p er cycle [22, 24]. F or later reference, the total energy released in A TP hydrolysis is approximately 20 k B T at ro om temp erature[27]. W e note that previ- ous work inv estigating trade-offs b et w een accuracy and energy in Mark ov chains used a non-thermodynamically feasible energy [28]. Our goal is to find the b est p erforming estimator for a giv en receptor arc hitecture and en tropy pro duction (en- ergy consumption) rate. Ho wev er, there are sev eral bio- logical constrain ts that need to b e considered when op- timizing ov er c hoices of kinetic parameters. First, the rate at which a chemical ligand binds to a receptor is set by diffusion limited binding [1] and hence k 01 is not con trolled b y the cell. Therefore w e set k 01 = 1 and do not optimize ov er this rate. Second, a receptor needs to b e specific. In principle, b oth “correct” ligands (i.e. the ligands the receptor has ev olved to detect) and “wrong” ligands (an y other c hemical) can bind the receptor. How- ev er, nonsp ecific ligands quickly unbind and cause the receptor to switch back to the nonsignaling state. Thus, the specificity of a receptor is set b y the mean duration of the signaling state in the presence of the correct ligand, τ S . This is incorp orated by requiring a small nonsp ecific binding rate ( k 0 L = 1 = k 01 ) and we do not opti- mize o ver k 0 L . Lastly , since any statistical estimator is alw ays improv ed with more samples, to fairly compare differen t families of estimates, we will fix the sampling rate, n = N /T , where N is the exp ected num b er of sam- ples and T is the signal in tegration time. By fixing the nonsp ecific binding rate ( k 0 L ) to b e small [24], this im- plies τ S ≈ n − 1 − 1. But since we are also fixing the sampling rate, n , this fixes τ S . In summary , our goal is to find the global minima for uncertaint y , giv en the abov e constrain ts. W e b egin b y analyzing the three-state receptor (Fig. 1F). Figure 2 sho ws the uncertain ty as a function of en tropy production for the optimal threeistate recep- tor for four different c hoices of the ligand binding rate, ¯ n = N /T . T o generate these plots, w e hav e used an analytic ansatz[24] for the optimal parameters which w e ha ve chec ked using sim ulated annealing (with agreement within 1 . 25%). Notice that learning more accurately (re- ducing uncertaint y) alw a ys increased energy consump- tion (entrop y pro duction). A t low energy consump- tion, the receptor approaches the equilibrium BP limit ( E = 2), while at high energy consumption (corresp ond- ing approximately to the energy of A TP hydrolysis) the optimal p erformance asymptotically approaches the infi- nite en trop y production analytic limit of h ( δ c ) 2 i c 2 ∼ 3 2 N (7) One striking observ ation is that these curv es exhibit a data collapse when plotted as a function of the energy consumption p er ligand binding rate, e p /n . The inset of Fig. 2 shows the same curves as the main graph as a function of e p /n . Since eac h ligand binding even t can b e viewed as an indep enden t sample of the external con- 4 cen tration, this data collapse suggests that the natural v ariable linking thermo dynamics and inference is the en- ergy p er indep enden t sample consumed in constructing an estimator. The three-state receptor is not able to reac h the MLE limit of E = 1 for any level of entrop y pro duction. T o reac h the MLE limit, w e consider a receptor with L + 1 states, L of which are signaling states (see Fig. 1G). This Marko v chain has 2 L indep endent parameters, whic h makes it hard to find the global optimum. F or this reason, we analyzed a simplified, but still biophys- ically realistic, rate structure (without p erforming any optimization ov er parameters) where k 01 , k 0 L , k 10 , k L 0 can indep enden tly v ary but all other forward rates are fixed to b e iden tical, k i,i +1 = f and all other bac kw ard rates chosen so that k i +1 ,i = b , where i = 1 . . . L − 1 [24]. Once again, for all choices of L , the optimal uncer- tain ty exhibits a data collapse as a function of the energy consumption p er ligand binding rate, e p / n (see Fig. 3). A t low energy consumption, the uncertaint y approaches the BP limit ( E = 2), while at high energy consumption (corresp onding appro ximately to the energy of A TP hy- drolysis) asymptotically approac hes the infinite entrop y pro duction analytic limit of h ( δ c ) 2 i c 2 ∼ 1 + 1 L 1 N (8) Th us, receptors with large energy consumption and man y signaling states ( L 1) approac h the MLE limit. In or- Entropy Production (e p ) Estimator ( E ) e p / n 1 A TP 1 A TP FIG. 2: Tw o signaling state estimator p erformance. F or v arying sampling rate n = N /T , the plot shows estimator p erformance ( E ) versus entrop y production ( e p with units of k B T = 1). The sym b ols represent results from simulated an- nealing, where k 01 = 1 and k 02 = = 10 − 3 while the other four rates are optimized. The con tinuous lines represen t our ansatz[24] for the global minima. At high en tropy production the estimators asymptotically approach 1 . 5. The inset shows the data collapse when the estimator p erformance ( E ) is plot- ted versus entrop y production p er sampling rate ( e p / n ). The v ertical dashed line corresp onds to the approximate energy released by h ydrolysis of a single A TP . 1.0 10 100 1.0 1.5 2.0 1 A TP 2 A TP L 3 9 27 Entropy Production / Sampling Rate ( e p / n ) Estimator ( E ) FIG. 3: Illustrative example of L signaling state estimator p erformance. F or a v arying n um b er of signaling states L , the plot sho ws estimator p erformance ( E ) v ersus energy consump- tion ( e p /n ). F or an increasing L , at high energy consumption the estimator approaches the maximum lik elihoo d limit of 1. The following parameters are fixed at n = 0 . 99, k 01 = 1, k 0 L = 10 − 3 , α = k 10 /b = 10 − 3 , and ω = k L 0 /f = 1, while b w as v aried to keep n fixed, and θ = f /b was v aried to c hange the estimator and the energy consumption. These parame- ters were c hosen for conv enience and are not global optima. The vertical lines correspond to the appro ximate energy re- leased b y hydrolysis of a single A TP (dashes) or tw o A TPs (dot dashes). der to p erfectly achiev e the MLE limit, all backw ard rates b would need to b e 0, leading to infinite entrop y pro duc- tion. An interesting feature of these curv es is that b e- y ond some scale (whic h can be ac hiev ed b y h ydrolysis of only a few A TP), the marginal gain in improv ement that results from consuming more energy b ecomes negligible. This is reminiscent of the recently found transition in ki- netic pro ofreading where adding additional energy only marginally improv es the error threshold [23, 29]. It will b e interesting to see if this is a generic feature of many bio c hemical information pro cessing circuits. In conclusion, b y analyzing the ability of cells to es- timate the concentration of an external c hemical signal using nonequilibrium receptors we hav e established an unexp ected link b et ween statistical inference and ther- mo dynamics. Sp ecifically , w e found that the efficacy of an estimator for the concen tration of a ligand dep ends on the energy consumed p er indep enden t sample by the receptor. Extrapolating this result suggests that there ma y b e fundamen tal thermo dynamic b ounds on statisti- cal inference. The trade-off betw een accuracy and energy is general and may b e relev ant for other signal transduc- tion systems, such as gene regulation [30], ligh t-activ ated proteins [31] or ligand-gated ion channels [32]. W e note that follo wing the tradition of Berg and Purcell, in this pap er w e only considered estimating a concen tration af- ter a long time T . How ever, in man y related cases, suc h as transcription [33], the sp eed is an imp ortant trade- 5 off in addition to accuracy and energy consumption. In the con text of phosphorelays, it is likely that the circuits can resp ond quic kly even for m ultistep cascades. F or example, the four-stage phosphorelay utilized for pho- totransduction in the retina can still respond to stim uli in ab out half a second [34]. Nonetheless, understanding these trade-offs represents an imp ortan t future research direction. W e conjecture that our observ ed scaling, ( e p /n ), re- flects a general principle: the efficiency of a statistical estimator is limited b y the energy consumed per sample during its construction. Of course, muc h more inv esti- gation is needed to see if this conjecture holds in gen- eral. In particular, it will b e interesting to see if these results change for receptors mo deled b y heterogeneous Mark ov netw orks that are not strictly ringlik e in nature. Recen t work indicates that at large en tropy production the dynamics of suc h netw orks may b e indep endent of de- tails of the underlying top ology , suggesting that our basic picture should hold ev en for more complicated nonequi- librium receptors [35]. An additional extension to our mo del would b e to consider externally v arying concentra- tions b y implementing a sensory adaptiv e system (SAS) as was done in recen t pap ers[36, 37]. These pap ers found that the accuracy and energy consumption of the SAS dep ends on the time scale of external concentration fluc- tuations. Finally , it is well known that many receptors, suc h as GPCRs, actively consume energy in order to op- erate. Our mo del presents one possible explanation for this observ ation. The energy consumption ma y help re- duce noise in the downstream signal, allowing cells to more accurately determine external concentrations. Our mo del also shows that h ydrolysis of only one or tw o A TP nearly ac hieves the theoretical minima of uncertain t y . This ma y explain why cell sensors often require only a few phosphorylation sites. W e w ould lik e to thank David Sch wab and Ja v ad No or- bakhsh for the useful discussions. W e also thank Luca D’Alessio and DJ Strouse for detailed commen ts on the pap er. AHL w as supported by a National Science F oun- dation Graduate Research F ellowship (NSF GRFP) un- der Gran t No. DGE-1247312. PM and CKF were sup- p orted b y a Sloan Research F ello wship and a NIH K25 GM086909. [1] H. Berg and E. Purcell, Bioph ys. J. 20 , 193 (1977). [2] W. Bialek and S. Setay eshgar, Pro c. Natl. Acad. Sci. U.S.A. 102 , 10040 (2005). [3] R. Endres and N. Wingreen, Phys. Rev. Lett. 103 , 158101 (2009). [4] B. Hu, W. Chen, W. Rapp el, and H. Levine, Phys. Rev. Lett. 105 , 48104 (2010). [5] T. Mora and N. Wingreen, Ph ys. Rev. Lett. 104 , 248101 (2010). [6] V. Sourjik and N. S. Wingreen, Curr. Opin. Cell Bio. 24 , 262 (2012). [7] K. Kaizu, W. de Ronde, J. P aijmans, K. T ak ahashi, F. T ostevin, and P . R. ten W olde, Biophys. J. 106 , 976 (2014). [8] R. Landauer, IBM J. Res. Dev. 5 , 183 (1961). [9] S. Laughlin, Curr. Opin. Neurobiol. 11 , 475 (2001). [10] P . Meh ta and D. J. Sch wab, Proc. Natl. Acad. Sci. U.S.A. 109 , 17978 (2012). [11] G. Lan, P . Sartori, S. Neumann, V. Sourjik, and Y. T u, Nat. Phys. 8 , 422 (2012). [12] C. C. Gov ern and P . R. ten W olde, Phys. Rev. Lett. 109 , 218103 (2012). [13] C. C. Gov ern and P . R. ten W olde, (2013). [14] A. Barato, D. Hartich, and U. Seifert, Ph ys. Rev. E 87 , 042104 (2013). [15] A. Berut, A. Arakely an, A. Petrosy an, S. Cilib erto, R. Dillensc hneider, and E. Lutz, Nature 483 , 187 (2012). [16] D. Mandal and C. Jarzynski, Pro c. Natl. Acad. Sci. U.S.A. 109 , 11641 (2012). [17] S. V aikuntanathan and C. Jarzynski, Phys. Rev. E 83 , 061120 (2011). [18] T. Saga wa and M. Ueda, Phys. Rev. E 85 , 021104 (2012). [19] S. Still, D. A. Siv ak, A. J. Bell, and G. E. Cro oks, Ph ys. Rev. Lett. 109 , 120604 (2012). [20] X. Cheng, L. Merc han, M. Tchernooko v, and I. Nemen- man, Phys. Bio. 10 , 035008 (2013). [21] J. E. Keymer, R. G. Endres, M. Skoge, Y. Meir, and N. S. Wingreen, Proc. Natl. Acad. Sci. U.S.A. 103 , 1786 (2006). [22] T.L. Hill, F r e e ener gy tr ansduction and bio chemical cycle kinetics , (Springer, 1989). [23] A. Murugan, D. A. Huse, and S. Leibler, Pro c. Natl. Acad. Sci. U.S.A. 109 , 12034 (2012). [24] See Supplementary Material for additional details. [25] G. Bel, B. Munsky , and I. Nemenman, Phys. Bio. 7 , 016003 (2010). [26] J. Leb owitz and H. Sp ohn, J. Stat. Phys. 95 , 333 (1999). [27] D. V o et and J. V o et, Bio chemistry (John Wiley and Sons New Y ork, 2004), 3rd ed. (pg 566, T able 16.3) [28] S. Escola, M. Eisele, K. Miller, and L. Paninski, Neural computation 21 , 1863 (2009). [29] B. Munsky , I. Nemenman, and G. Bel, The J. Chem. Ph ys 131 , (2009). [30] D. M. Suter, N. Molina, D. Gatfield, K. Schneider, U. Schibler, and F. Naef, Science 332 , 472 (2011). [31] W. Bialek, Biophysics: Se ar ching for Principles (Prince- ton Universit y Press, 2012). [32] L. Csan´ ady , P . V ergani, and D. C. Gadsby , Proc. Natl. Acad. Sci. U.S.A. 107 , 1241 (2010). [33] M. Depken, J. M. Parrondo, and S. W. Grill, Cell Rep orts 5 , 521 (2013). [34] P . B. Detwiler, S. Ramanathan, A. Sengupta, and B. I. Shraiman, Biophysical Journal 79 , 2801 (2000). [35] S. V aikuntanathan, T. R. Gingric h, and P . L. Geissler, Ph ys. Rev. E 89 , 062108 (2014). [36] P . Sartori, L. Granger, C. F. Lee, and J. M. Horowitz, arXiv:1404.1027 (2014). [37] A. C. Barato, D. Hartich, and U. Seifert, (2014). [38] S. Redner, A Guide to First-Passage Pr o c esses (Cam- bridge, 2001). [39] R. A. Blythe, Master’s thesis, Edin burgh (2001). 6 [40] F. Hussain, C. Gupta, A. J. Hirning, W. Ott, K. S. Matthews, K. Josi ´ c, and M. R. Bennett, Pro ceedings of the National Academy of Sciences 111 , 972 (2014). [41] I. H. Segel, Enzyme Kinetics , vol. 360 (Wiley , New Y ork, 1975). Supplemen tary Material Notation Here we pro vide more details of the results in the main text. First, w e outline our notation. The time dependent probabilit y of state i is p i = p i ( t ), while the steady state probability of state i is p ss i . The Laplace transformed probabilit y of state i is P i ( s ). The rate to go from state i to state j is k ij . The probability to transition from state i to state j is q ij . The time it tak es to transition from state i to j is τ ij . The first passage time is given b y f ( t ) while the Laplace transformed first passage time is F ( s ). The lifetime of state i is ρ i . Detailed Deriv ation of General Uncertain t y Here w e deriv e form ulas for the accuracy of statistical inference when the activ ated signaling states contin uously pro duce signals. F ollowing Berg and Purcell [1], w e will measure the accuracy of a receptor by the “uncertain t y” of the concen tration estimate: uncertain ty := h ( δ c ) 2 i c 2 (9) where c is the mean and h ( δ c ) 2 i is the v ariance of the estimated concen tration. Let us consider the case where activ ated signaling states pro duce do wnstream signaling molecules at a rate α . W e will define τ S as the mean lifetime of the signaling states and τ N S as the mean non-signaling time. Then, we kno w that the mean n umber of signaling molecules u pro duced after a time T is given by u = αT τ S τ S + τ N S ≡ αT ¯ φ (10) This follows b y noting that ¯ φ is just the fraction of time the receptor is in the signaling states. Notice that by definition, α and T are indep endent of the concen tration c . The signaling time τ S , can in principle dep end on concen tration, and for L signaling states is giv en b y τ S = L X i =1 p 0 i τ i 0 (11) where p 0 i is the probabilit y to transition from state 0 to state i , and τ i 0 is the mean time to return from state i to state 0. Since w e assume the receiving state is strongly biased (i.e. k 01 is muc h larger than any other rate k 0 i from non-signaling, 0, to signaling state i ), then the deriv ativ e of the signaling time with resp ect to concentration is: dτ S dc = − L X i =2 k 0 i k 01 ( τ 10 + τ i 0 ) (12) Since this is b y assumption small, we will approximate τ S as independent of concen tration, and thus all the concen- tration dependence comes from τ N S . Thus, using the usual error-propagation formulas one has δ u u = − dτ N S dc 1 τ S + τ N S δ c (13) whic h giv es the uncertaint y for the concen tration: h ( δ c ) 2 i c 2 = c dτ N S dc − 2 ( τ N S + τ S ) 2 h ( δ u ) 2 i u 2 (14) 7 The formula ab o ve reduces the problem to calculating the uncertaint y in the num b er of signaling molecules pro duced in a time T . T o calculate this, notice that u comes from on av erage N = T / ( τ S + τ N S ) indep enden t binding cycles (state 0 to state 1 transition). Th us, the v ariance in the fraction of time bound during a time T will just b e N − 1 times the v ariance in a single binding cycle. In particular, the co efficien t of v ariation in a single cycle is giv en b y δ φ φ = τ N S τ S + τ N S δ τ S τ S − δ τ N S τ N S (15) Noting that the signaling and non-signaling ev ents are independent, w e get h ( δ u ) 2 i u 2 = 1 N τ N S τ S + τ N S 2 h ( δ τ N S ) 2 i τ 2 N S + h ( δ τ S ) 2 i τ 2 S (16) Plugging this expressions in to (14) gives h ( δ c ) 2 i c 2 = 1 N c d log ( τ N S ) dc − 2 h ( δ τ N S ) 2 i τ 2 N S + h ( δ τ S ) 2 i τ 2 S (17) Therefore the complicated resp onse of a receptor is reduced to its mean and v ariance of the time in b oth the signaling and non-signaling states. In this pap er, we will examine the case where there is a single non-signaling state (0) and there are L signaling states arranged in a ring. In this case, the ab ov e expression simplifies to (leading order k 0 L /k 01 ): h ( δ c ) 2 i c 2 = 1 N 1 + h ( δ τ S ) 2 i τ 2 S (18) F or a tw o state pro cess as considered by Mora and Wingreen [5], there is only the receiving state and one signaling state. These are just P oisson processes which eac h ha ve an uncertaint y of 1 and w e recov er the Berg and Purcell [1] limit h ( δ c ) 2 i c 2 = 2 N (19) General First Passage Time W e need to calculate the first passage prop erties of the Marko v chain, specifically the mean and v ariance of the first passage time. This can b e calculated as follows [25, 38]. The master equation that w e wan t to solv e is dp dt = K p ( t ). First apply the Laplace transform P i ( s ) = Z ∞ 0 p i ( t ) e − st dt (20) whic h leads to the master equation ( s − K ) P ( s ) = p ( t = 0) (21) with K the matrix of transitions for the full system but with the transition rates lea ving the absorbing states set to zero. The first passage time to return to state 0 is f ( t ) = dp 0 ( t ) dt (22) F ( s ) = sP 0 ( s ) (23) F or our purp oses, w e only need the mean and v ariance of the first passage time. This is easily obtained by the uncen tered momen ts M ( m ) = Z ∞ 0 t m f ( t ) = ( − 1) m d m F ( s ) ds m s =0 (24) 8 where m = 1 is the mean and m = 2 is the uncen tered second moment. In general w e kno w that τ x , the spent in state x , is dra wn from a mixture where it can switc h to states j = 1 , 2 , ... . The v ariance of mixtures is X = P i w i X i , where w i are arbitrary weigh ts and X i are random v ariables drawn from distributions with mean µ i and v ariance σ i . Combining equations we get: V ar(X) = X i w i ( µ i − µ ) 2 + σ 2 i (25) with µ = P i w i µ i . W e can get the time sp en t in state x , τ x , by using the v ariance mixture form ula combined with τ ix and V ar( τ ix ), resp ectiv ely the mean and v ariance first passage time of starting in state i and ending in state x . This giv es us τ x = X i q xi τ ix (26) q xi = k xi P j k xj = k xi ρ x (27) ρ x = X j k xj − 1 (28) V ar( τ x ) = X i q xi V ar ( τ ix ) + X i q xi τ ix − X k q xk τ kx ! 2 (29) where q xi is the probabilit y of transitioning from state x to state i , k xi is the rate to go from state x to state i , and ρ x is the lifetime of state x . In this pap er, w e hav e one non-signaling state and the other L states are signaling. Therefore, w e will let state 0 b e the absorbing state, and it can initially transition to state 1 and state L . The abov e equations then simplify to τ 0 = q 01 τ 10 + q 0 L τ L 0 (30) V ar( τ 0 ) = q 01 V ar ( τ 10 ) + q 0 L V ar ( τ L 0 ) + 2 q 01 q 0 L ( τ 10 − τ L 0 ) 2 (31) q 0 L = 1 − q 01 (32) First Passage Time: 2 Signaling States Here we calculate the mean and v ariance of the first passage time to return to state 0 from either state 1 or 2. The master equation that w e need to solve is dp dt = K p ( t ). The matrix rates are: K ij = k 10 for i = 0 and j = 1 k 12 for i = 2 and j = 1 k 20 for i = 0 and j = 2 k 21 for i = 1 and j = 2 − ( k 10 + k 12 ) for i = 1 and j = 1 − ( k 20 + k 21 ) for i = 2 and j = 2 0 ev erywhere else (33) While the initial conditions are set by the rates k 01 and k 02 , for the purp oses of the first passage time calculation, the rates from 0 to 1 ( k 01 ) and from 0 to 2 ( k 02 ) are b oth set to zero, k 01 = k 02 = 0. The Laplace transform for the initial condition of starting in state 1 is: F ( s ) = sP 0 ( s ) = k 10 P 1 + k 20 P 2 (34) 9 with P 1 = Γ 1 − k 12 k 21 Γ 2 − 1 (35) P 2 = k 12 Γ 2 P 1 (36) Γ i = s + ρ − 1 i (37) W e can obtain mean and v ariance from τ = − dF ds s =0 (38) V ar( τ ) = d 2 F ds 2 s =0 − τ 2 (39) The mean and v ariance of the first passage time from starting in either state 1 or state 2 is: τ 10 = ρ 1 1 + k 12 ρ 2 1 − k 12 k 21 ρ 1 ρ 2 = k 12 + k 20 + k 21 ξ (40) τ 20 = ρ 2 1 + k 21 ρ 1 1 − k 12 k 21 ρ 1 ρ 2 = k 10 + k 12 + k 21 ξ (41) V ar( τ 10 ) = τ 2 10 1 + 2 ρ 2 2 k 12 ( k 10 − k 20 ) (1 + k 12 ρ 2 ) 2 = τ 2 10 + 2 k 12 ( k 10 − k 20 ) ξ 2 (42) V ar( τ 20 ) = τ 2 20 1 + 2 ρ 2 1 k 21 ( k 20 − k 10 ) (1 + k 21 ρ 1 ) 2 = τ 2 20 − 2 k 21 ( k 10 − k 20 ) ξ 2 (43) ξ = k 10 k 20 + k 10 k 21 + k 12 k 20 (44) where the second equalit y holds as long as ξ 6 = 0. First Passage Time: L Signaling States Deriv ation Here we calculate the mean and v ariance of the first passage time in a L + 1 state c hain. The master equation that w e need to solve is dp dt = K p ( t ). The matrix is indexed from 0 to L and the rates are: K ij = k 10 for i = 0 and j = 1 k L 0 for i = 0 and j = L f for i = j + 1 and 1 < j < L b for i = j − 1 and 1 < j < L − ( f + k 10 ) for i = 1 and j = 1 − ( f + b ) for i = j and 1 < j < L − ( k L 0 + b ) for i = L and j = L 0 ev erywhere else (45) While the initial conditions are set by the rates k 01 and k 0 L , for the purposes of the first passage time calculation, the rates from 0 to 1 ( k 01 ) and from 0 to L ( k 0 L ) are b oth set to zero, k 01 = k 0 L = 0. F or later con venience we define the follo wing ratio of rates: θ = f b (46) α = k 10 b (47) ω = k L 0 f (48) 10 W e can use a transfer matrix to find a general solution (for non-degenerate eigenv ales, i.e. θ 6 = 1) to the state probabilit y as P i ( s ) = C + λ i − 1 + + C − λ i − 1 − (49) Solving for the the expressions 1 < i < L leads to λ ± = 1 2 b s + f + b ± p ( s + f + b ) 2 − 4 f b (50) = 1 2 σ ± p σ 2 − 4 θ = 1 2 ( σ ± ψ ) (51) σ = s b + θ + 1 (52) ψ = p σ 2 − 4 θ (53) With the initial condition of starting in P 1 , the b oundary equations for P 1 and P L are: ( σ + α − 1) ( C + + C − ) = 1 /b + ( C + λ + + C − λ − ) (54) ( σ + ( ω − 1) θ ) C + λ L − 1 + + C − λ L − 1 − = θ C + λ L − 2 + + C − λ L − 2 − (55) Solving these equations giv es C − = − C + Λ L M (56) C + = 1 b [ λ − + α − 1 − ( λ + + α − 1)Λ L M ] (57) Λ = λ + λ − (58) M = 1 + ( ω − 1) λ − 1 + ( ω − 1) λ + (59) And then the probabilities are P 1 ( s ) = C + 1 − Λ L M (60) P L ( s ) = C + λ L − 1 + (1 − Λ M ) (61) signal signal signal 0 1 2 L k 10 k 01 b f k 0L k L0 FIG. 4: Simplified rate structure considered for L signaling states first passage time calculation. The rates k 01 , k 10 , k L 0 , k 0 L are unconstrained, while the remaining forward rates are equal, f = k 12 = k 23 = . . . = k L − 1 ,L and the remaining bac kw ard rates are equal, b = k 21 = k 32 = . . . = k L,L − 1 . 11 The full Laplace transform F is: F ( s ) = α (1 − Λ L M ) + ω θ λ L − 1 + (1 − Λ M ) λ − + α − 1 − ( λ + + α − 1)Λ L M (62) Results T o get the mean and v ariance of the first passage time, we need τ 10 = − dF ds s =0 (63) V ar( τ 10 ) = d 2 F ds 2 s =0 − τ 2 (64) The mean return time to state 0 when starting in state 1 is: τ 10 = τ 10 ,num τ 10 ,den (65) τ 10 ,num = ( ω L − ω + 1) θ L +1 − ( ω L + 1) θ L + ( ω − 1) θ + 1 (66) τ 10 ,den = b [ θ − 1] ω θ L +1 + ω ( α − 1) θ L + α (1 − ω ) θ − α (67) The v ariance of the return time to state 0 when starting in state 1 is: V ar( τ 10 ) = V ar( τ 10 ) num V ar( τ 10 ) den (68) V ar( τ 10 ) num = θ 2 L +3 ω 2 ( L − 1) + 1 (69) + θ 2 L +2 ω 2 L 2 α − L (3 α + 1) + 2 α − 3 + 2 ω (( L − 2) α + 1) + 2 α − 3 − θ 2 L +1 ω 2 2 L 2 α − 4 Lα + L + 4 α − 4 + ω ((4 L − 6) α + 4) + 4 α − 3 + θ 2 L [ ω ( ω L + 2)( Lα − α + 1) + 2 α − 1] + θ L +3 ( ω − 1) 2( ω − 1) α + 3 ω L 2 α + L ( ω (4 − 5 α ) + 4 α ) + 2 + θ L +2 − 2 ω 2 3 L 2 α + L (4 − 6 α ) + α − 2 + θ L +2 ω 9 L 2 α + L (12 − 23 α ) + 8 α − 6 + 6(2 L − 1) α + 6 + θ L +1 ω − 9 L 2 α + L (19 α − 12) − 6 α + 6 + θ L +1 ω 2 ( L − 1)((3 L − 4) α + 4) + 6( − 2 Lα + α − 1) + θ L α 3 L 2 ω − 5 Lω + 4 L + 2 ω − 2 + (4 L − 2) ω + 2 − θ 3 ( ω − 1) 2 (2 α − 1) − θ 2 ( ω − 1)( ω + 4 α − 3) + θ ( − 2 ω − 2 α + 3) − 1 V ar( τ 10 ) den = b 2 [ θ − 1] 3 ω θ L +1 + ω ( α − 1) θ L + α (1 − ω ) θ − α 2 (70) While the results here are for initial condition of b eing in state 1, one can easily find the results for the initial condition of state L if one mak es the following substitutions θ ⇔ 1 /θ , b ⇔ f , and α ⇔ ω . Steady State Probabilities In general, we are considering a Marko v c hain with L + 1 nodes (lab eled 0 to L ). W e ha ve the master equation dP ( t ) dt = K P ( t ) (71) 12 with K the matrix of transition rates. The rates are lab eled as k ij where i is the initial state and j is the final state. F or later con venience, define the lifetime of a state as ρ i = X j 6 = i k ij − 1 (72) The steady state distributions are easily obtained by solving K p ss = 0. The solution can b e written in a compact form [39] as P ss i = z i Z (73) Z = X i z i (74) and z i is the matrix minor of K at ( i, i ) i.e. the determinan t of K with the i th row and column remov ed. F or the t wo signaling state system we ha ve that p ss 0 = ρ − 1 1 ρ − 1 2 − k 12 k 21 Z = k 10 k 20 + k 10 k 21 + k 12 k 20 Z (75) p ss 1 = ρ − 1 0 ρ − 1 2 − k 02 k 20 Z = k 01 k 20 + k 01 k 21 + k 02 k 21 Z (76) p ss 2 = ρ − 1 0 ρ − 1 1 − k 01 k 10 Z = k 01 k 12 + k 02 k 10 + k 02 k 12 Z (77) Z = X i 6 = j ρ − 1 i ρ − 1 j − k ij k j i (78) F or the L signaling state with the simplified rates, we will just presen t the result for state 0: p ss 0 = p ss 0 ,num p ss 0 ,den (79) p ss 0 ,num = b ( θ − 1) ω θ L +1 + ω ( α − 1) θ L + α (1 − ω ) θ − α (80) p ss 0 ,den = − α + αb + αL + + 1 (81) + θ ( αbω − 2 αb − αL + ω − − 1) (82) + αbθ 2 (1 − ω ) (83) + θ L ( bω + α − Lω − 1 − − αbω ) (84) + θ 1+ L ( αbω − 2 bω + Lω − ω + 1 + ) (85) + bω θ L +2 (86) The rates from 0 to 1 is k 01 = 1, from 1 to 0 is k 10 (with α = k 10 /b ), from 0 to L is k 0 L = 1, and from L to 0 is k L 0 (with ω = k L 0 /f ). All other forw ard rates are f and bac kward rates are b and the ratio of rates is θ = f /b . Av erage Sampling Rate: n The a v erage sampling rate is n = N T = k 01 p ss 0 (87) where N is the n umber of samples (i.e. num b er of binding even ts), T is the total in tegration time, k 01 is the rate from state 0 to state 1, and p ss 0 is the steady state probabilit y of being in state 0. Since we are assuming that k 01 = 1 and k L 0 = 1, we hav e the mean signaling time b ecomes τ S ≈ τ 10 . With these rates we hav e n ≈ (1 + τ S ) − 1 (88) 13 En tropy Production: e p F or a general Marko v process with states lab eled by i , steady state probabilities p ss i , and transition rate k ij from state i to state j , the non-equilibrium steady state (NESS) entrop y pro duction [10, 26] is given by e p = L X i =0 L X j 6 = i p ss i k ij ln k ij k j i (89) where the summation is o v er both i and j . Alternativ ely , the en tropy pro duction can b e written as a sum ov er the flux betw e en each connected no de as e p = L X i =0 L X j >i ( p ss i k ij − p ss i k ij ) ln k ij k j i (90) where no w we ha ve an unrestricted sum ov er i but a restricted sum ov er j . Since w e are mo deling our receptor as a ring, the en tropy production simplifies to e p = ( p ss 0 k 01 − p ss 1 k 10 ) ln k 01 k 12 . . . k L 0 k 0 L k 10 . . . k L,L − 1 = J ln γ (91) where the flux J = p ss 0 k 01 − p ss 1 k 10 b et w een each neigh b oring state is equal and the ln γ is the free energy difference of a cycle. F or 2 signaling states, the en tropy pro duction p er sampling rate is given by: e p n = 1 + k 10 k 12 + k 10 k 21 k 12 k 20 − 1 γ − 1 γ ln γ (92) γ = k 01 k 12 k 20 k 10 k 21 k 02 (93) F or the L signaling states arranged in a ring, the en tropy pro duction per sampling rate is giv en b y: e p n = 1 + α ω θ − L + α θ − 1 1 − θ 1 − L 1 − θ − 1 − 1 γ − 1 γ ln γ (94) γ = k 01 ω k 0 L α θ L (95) where ω = k L 0 /f , α = k 10 /b , θ = f /b , f is all the forw ard rates (except k 01 and k L 0 ), and b is all the bac kward rates (except k 10 and k 0 L ). Ansatz for 2 Signaling State Receptor Here are the details of the ansatz for the minim um uncertain ty for the 2 signaling state system. The rates are as follo ws: • k 01 = 1 • k 10 = k 2 (1 − x ) • k 12 = k x • k 21 = k δ • k 20 = k • k 02 = 14 where 1 (and in this pap er = 10 − 3 ), 0 < x < 1, δ 1 (and in this paper δ = 0 . 04), and k is v aried to fix the mean sampling rate n . F or the ansatz, the mean, coefficient of v ariation, and entrop y production simplifies to τ S ≈ 2 k (96) h ( δ τ S ) 2 i τ 2 S ≈ 1 − x 1 + x (97) e p n ≈ 1 + 1 − x 2 x − 1 γ − 1 γ ln γ (98) γ = 2 x δ (1 − x ) (99) Sim ulated Annealing Sim ulated annealing is a meta-heuristic algorithm for global optimization in which one uses the Metrop olis algorithm to p erform a random walk in parameter space while p erio dically low ering the temp erature. W e used a simulated annealing algorithm to search for the parameters of a model describing a receptor with 2 signaling states that minimizes a cost function giv en b y cost = h ( δ c ) 2 i ¯ c 2 + λ e p (ln e p − ln ˆ e p ) 2 − ( λ n ˆ n − 1) ln n − ( λ n (1 − ˆ n ) − 1) ln(1 − n ) (100) That is, we minimize the uncertaint y of the resulting estimator ( h ( δ c ) 2 i / ¯ c 2 ) sub ject to soft constrain ts on the energy pro duction ( e p ) and sampling rate ( n ), which are constrained to ˆ e p and ˆ n , resp ectively . Here, λ e p and λ n implemen t the constrain ts. W e c hose λ e p = 20 and λ n = 20 / max { ˆ n, 1 − ˆ n } . Let Ω 1 denote a set of parameters describing a receptor with 2 signaling states (i.e. all of the v arious rate constan ts). A new set of trial parameters Ω 2 w as generated in the follo wing w ay: for eac h k ∈ Ω 1 set the corresponding k 0 ∈ Ω 2 to ln k 0 = ln k + η where η is a random v ariable with from a Normal distribution cen tered at zero. The width of the signal signal 0 1 2 k 10 k 10 = k(1-x)/2 1 k δ kx ε k FIG. 5: Rate structure for ansatz of minim um uncertain ty for the L = 2 signaling state system. The rates are as follo ws: k 01 = 1, k 10 = k 2 (1 − x ), k 12 = kx , k 21 = kδ , k 20 = k , and k 02 = . The mean signaling time is set b y k . The other rates are , δ 1 and 0 < x < 1. 15 Normal distribution w as chosen adaptively so that approximately 25% of the steps were accepted. Making the random p erturbations to the logarithm of the rate constants ensures that they are alw ays p ositive. The trial mo v e was accepted according to the Metropolis criterion with probability min[1 , exp((cost(Ω 1 ) − cost(Ω 2 )) /T )]. The temperature T w as initialized to T = 10 and adjusted b y T ← 0 . 95 T every 2000 steps. The b est solution obtained during the chain was stored in Ω B , and the chain was re-initialized from Ω 1 = Ω B ev ery 2000 steps to preven t the chain from getting stuck in a p o or local minim um. This sim ulated annealing algorithm was run until conv ergence of h ( δ c ) 2 i / ¯ c 2 , e p and n . Scaling with T emp erature In the main text, we work ed in the units of k B T = 1. How ev er, here we examine the general temp erature dep endence. Exp erimen tally , it is known that rates of biochemical reactions doubles for ev ery 10 ◦ C [40, 41]. Therefore, a general rate k at a temp erature T (measured in degrees Celsius) is related to initial rate k 0 and initial temp erature T 0 b y: k = k 0 2 T − T 0 10 (101) No w we need to determine the general scaling of v arious entities in this pap er, which is summarized b elow in terms of a general rate k : • Mean signaling time, τ S ∼ k − 1 • V ariance in signaling time, h ( δ τ S ) 2 i ∼ k − 2 • Co efficien t of v ariation of signaling time, h ( δ τ S ) 2 i τ 2 S ∼ 1 • Sampling rate, n ∼ k • Uncertainity , h ( δ c ) 2 i c 2 ∼ k − 1 • Entrop y pro duction, e p ∼ k While increasing temp erature increases b oth the mean and v ariance of the signaling time, since the estimator ( E = 1 + h ( δ τ S ) 2 i τ 2 S ) only dep ends on the co efficien t of v ariation of signaling time, the estimator is indep enden t of temp erature. The sampling rate n does increase with increasing temperature, and therefore increasing temp erature decreases the uncertaint y . How ev er, this decrease in uncertaint y costs energy . While the free energy p er cycle (ln γ ) remains constant, the probability flux ( J ) is prop ortional to a rate, and since the entrop y pro duction is giv en by e p = J ln γ , w e see that that decrease in uncertaint y is directly related to the increase in en trop y production.
Original Paper
Loading high-quality paper...
Comments & Academic Discussion
Loading comments...
Leave a Comment