Remodeling of Fibrous Extracellular Matrices by Contractile Cells: Predictions from Discrete Fiber Network Simulations
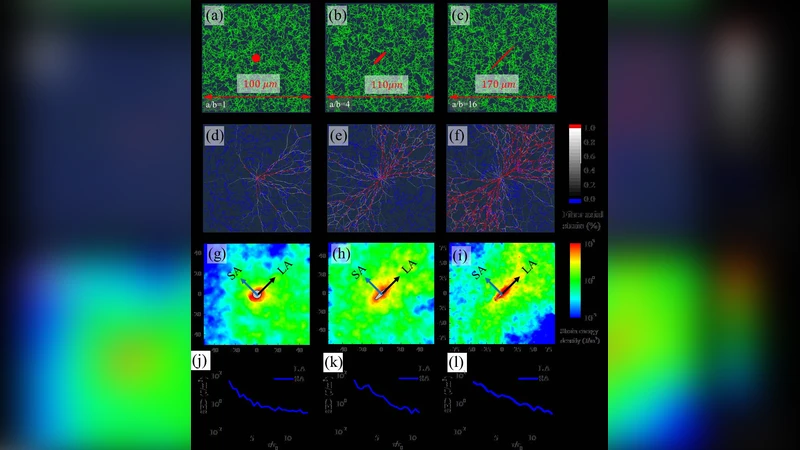
Contractile forces exerted on the surrounding extracellular matrix (ECM) lead to the alignment and stretching of constituent fibers within the vicinity of cells. As a consequence, the matrix reorganizes to form thick bundles of aligned fibers that enable force transmission over distances larger than the size of the cells. Contractile force-mediated remodeling of ECM fibers has bearing on a number of physiologic and pathophysiologic phenomena. In this work, we present a computational model to capture cell-mediated remodeling within fibrous matrices using finite element based discrete fiber network simulations. The model is shown to accurately capture collagen alignment, heterogeneous deformations, and long-range force transmission observed experimentally. The zone of mechanical influence surrounding a single contractile cell and the interaction between two cells are predicted from the strain-induced alignment of fibers. Through parametric studies, the effect of cell contractility and cell shape anisotropy on matrix remodeling and force transmission are quantified and summarized in a phase diagram. For highly contractile and elongated cells, we find a sensing distance that is ten times the cell size, in agreement with experimental observations.
💡 Research Summary
This paper presents a computational framework that captures how contractile cells remodel fibrous extracellular matrices (ECM) by aligning and stretching collagen fibers. The authors employ a finite‑element (FE) based discrete fiber network (DFN) model in which each collagen fiber is represented as an individual nonlinear truss element with a strain‑dependent tension‑stretch relationship. A random three‑dimensional fiber mesh is generated according to experimentally measured distributions of fiber length, diameter, and connectivity. Cells are introduced as geometric inclusions (circular or elliptical) that impose a uniform isotropic contractile stress σc on the surrounding matrix. Cell shape anisotropy is quantified by the aspect ratio α (length/width). By varying σc, α, and cell size R, the authors explore how these parameters control matrix deformation, fiber alignment, and the spatial extent of force transmission.
Key findings include: (1) Contractile cells induce radial alignment of fibers, with a pronounced bundle forming along the cell’s long axis. The degree of alignment, measured by an orientation order parameter S, increases sharply when the product σc·α exceeds a critical threshold. (2) Aligned bundles act as stiff “highways” that convey tensile stresses far beyond the cell’s physical dimensions. The authors define a sensing distance Ls as the radius at which matrix strain falls below a small reference value; for highly contractile, elongated cells, Ls can reach ~10 R, matching experimental observations of long‑range mechanosensing. (3) When two cells are placed within each other’s sensing zones, the bundles can merge, leading to cooperative force transmission if the bundles are co‑oriented, or to mechanical isolation if they are orthogonal. This provides a mechanistic explanation for cell‑cell mechanical communication in dense tissues. (4) A parametric phase diagram is constructed in the σc–α space, delineating regimes of weak remodeling (low σc·α) and strong remodeling (high σc·α) characterized by abrupt transitions in S and Ls. The diagram offers a predictive tool for assessing how different cell types (e.g., fibroblasts, myofibroblasts, cancer cells) remodel their microenvironment.
The model reproduces several experimental phenomena: heterogeneous strain fields around single cells, formation of thick, aligned collagen bundles, and the ability of a single cell to influence matrix mechanics over distances an order of magnitude larger than its own size. By explicitly modeling individual fibers, the DFN approach captures the non‑affine deformations and strain‑induced reorientation that are lost in continuum descriptions.
Limitations are acknowledged. Fiber‑to‑fiber shear interactions and cross‑link dynamics are simplified, and the intracellular contractile machinery is reduced to a uniform contractile stress rather than a detailed actomyosin network. Consequently, the model does not yet account for dynamic matrix remodeling processes such as fiber synthesis, degradation, or enzymatic cross‑linking. Future extensions could incorporate viscoelastic fiber behavior, active remodeling rules, and feedback between cell signaling and matrix mechanics.
In summary, this study provides a robust, physics‑based simulation platform that links cellular contractility and geometry to ECM fiber alignment, heterogeneous deformation, and long‑range force transmission. The quantitative phase diagram and the demonstrated ability to predict single‑cell and pairwise interactions make the work highly relevant for tissue engineering, wound healing, and cancer metastasis research, where understanding and controlling cell‑matrix mechanical communication is essential.
Comments & Academic Discussion
Loading comments...
Leave a Comment