Kinesin Motor Transport is Altered by Macromolecular Crowding and Transiently Associated Microtubule-Associated Proteins
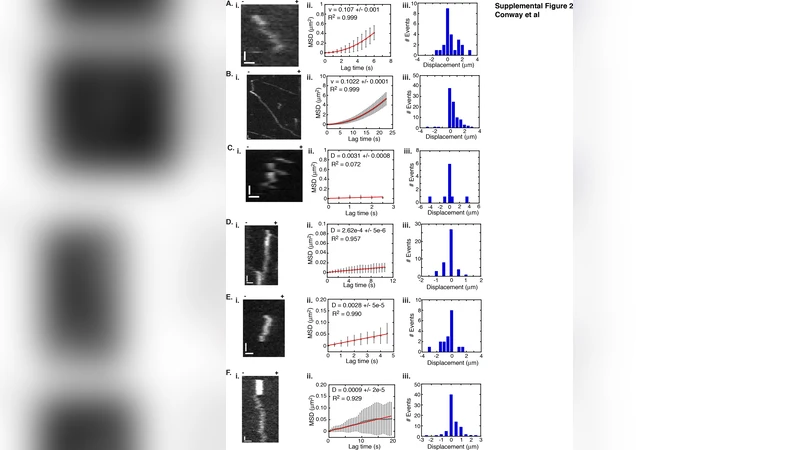
Intracellular transport of vesicular cargos, organelles, and other macromolecules is an essential process to move large items through a crowded, and inhomogeneous cellular environment. In an effort to dissect the fundamental effects of crowding and an increasingly complex cellular environment on the transport of individual motor proteins, we have performed in vitro reconstitution experiments with single kinesin-1 motors walking on microtubules in the presence of crowding agents and transient microtubule-associated proteins that more closely emulate the cellular environment. Macromolecular crowding due to inert polymers caused enhanced run lengths of motors, but displayed an increased tendency for non-specific motor association and diffusion, most likely due to depletion interactions. We found that transiently bound associated proteins slowed forward motion, but did not drastically affect the association times, in opposition to previously reported obstacle properties of stably associated microtubule-associated proteins, such as the neuronal protein tau. Such studies of the transport properties of molecular motors in increasingly complex reconstituted environments are important to illuminate the fundamental biophysical principles underlying the essential process of intracellular cargo transport.
💡 Research Summary
Intracellular cargo transport relies on the coordinated activity of microtubule‑based motors such as kinesin‑1. While most in‑vitro studies have examined motor behavior on pristine microtubules in dilute solutions, the cytoplasm is a highly crowded, heterogeneous medium populated by a variety of microtubule‑associated proteins (MAPs). This paper addresses the gap by reconstituting a more physiologically relevant environment: single kinesin‑1 molecules were observed moving on surface‑immobilized microtubules in the presence of (i) inert macromolecular crowding agents (polyethylene glycol, PEG) that mimic the excluded‑volume effects of the cytoplasm, and (ii) transiently binding MAPs (e.g., MAP65) that associate and dissociate on the millisecond‑second timescale, in contrast to the stable MAP tau.
Using total internal reflection fluorescence (TIRF) microscopy, the authors tracked hundreds of individual motor trajectories and extracted quantitative parameters: forward velocity, run length, dwell (association) time, the frequency of non‑specific binding events, and one‑dimensional diffusion coefficients. The key findings can be grouped into two themes.
First, macromolecular crowding dramatically enhances kinesin processivity. In 20 % w/v PEG, the average run length increased from ~1.2 µm (no crowding) to ~1.8 µm, and the dwell time on the microtubule was extended by ~1.5‑fold. This effect is attributed to depletion interactions: the polymer network reduces the effective volume available to the motor‑microtubule complex, thereby lowering the free‑energy barrier for staying attached. However, crowding also raises the incidence of non‑specific interactions; a subset of trajectories displayed diffusive, rather than directed, motion, indicating that the polymer “coat” can weaken the specific motor‑track binding energy and promote transient sliding.
Second, transient MAPs modulate motor speed without substantially altering attachment stability. Adding MAP65 at micromolar concentrations slowed kinesin’s average velocity by 20‑30 % (from ~0.8 µm s⁻¹ to 0.55‑0.65 µm s⁻¹) while leaving run length and dwell time essentially unchanged. The authors interpret this as a dynamic friction effect: MAPs briefly increase the local surface stickiness, causing a temporary drag on the motor. Because MAP65 rapidly dissociates (k_off on the order of seconds), the motor can resume its original speed once the obstacle disappears, unlike the permanent roadblock presented by tau, which dramatically reduces both run length and attachment probability.
These observations have several broader implications. The enhancement of processivity by crowding suggests that the dense cytoplasmic milieu may actually facilitate long‑range cargo delivery, counterbalancing the intuitive notion that crowding merely impedes motion. Conversely, the rise in non‑specific binding and diffusion highlights a trade‑off: increased processivity may come at the cost of reduced directional fidelity. The distinct influence of transient MAPs points to a cellular strategy for fine‑tuning transport rates without sacrificing motor engagement. By dynamically altering microtubule surface properties, cells could locally regulate cargo flux, for example, during axonal growth or synaptic remodeling.
Methodologically, the study demonstrates the value of adding two physiologically relevant parameters—crowding density (φ) and MAP kinetic rates (k_on, k_off)—to existing motor transport models. Incorporating these factors can improve predictions of cargo delivery times, spatial distribution, and the response to pathological alterations (e.g., tau aggregation in neurodegeneration).
Future directions proposed include (1) testing multiple motor types and cooperative ensembles, (2) mixing stable and transient MAPs to explore combinatorial effects, and (3) employing cell‑derived cytoplasmic extracts to capture the full spectrum of macromolecular interactions.
In summary, the paper provides compelling evidence that kinesin‑1 transport is not solely dictated by the intrinsic properties of the motor or the microtubule lattice, but is profoundly shaped by the surrounding macromolecular crowding and the dynamic landscape of associated proteins. These findings deepen our mechanistic understanding of intracellular logistics and offer new design principles for synthetic nanotransport systems and therapeutic strategies targeting transport defects.