Sitting at the edge: How biomolecules use hydrophobicity to tune their interactions and function
Water near hydrophobic surfaces is like that at a liquid-vapor interface, where fluctuations in water density are substantially enhanced compared to that in bulk water. Here we use molecular simulations with specialized sampling techniques to show that water density fluctuations are similarly enhanced, even near hydrophobic surfaces of complex biomolecules, situating them at the edge of a dewetting transition. Consequently, water near these surfaces is sensitive to subtle changes in surface conformation, topology, and chemistry, any of which can tip the balance towards or away from the wet state, and thus significantly alter biomolecular interactions and function. Our work also resolves the long-standing puzzle of why some biological surfaces dewet and other seemingly similar surfaces do not.
💡 Research Summary
This paper investigates how water behaves near hydrophobic regions of biomolecules and demonstrates that such water is poised at the edge of a dewetting transition, making it exquisitely sensitive to minute perturbations in surface geometry, chemistry, or external fields. Using molecular dynamics simulations combined with an advanced version of the Indirect Umbrella Sampling (INDUS) technique, the authors quantify water density fluctuations by measuring the probability distribution P v(N) of finding N water molecules inside arbitrarily shaped observation volumes v placed adjacent to the surface of interest.
The study begins with simple model systems: self‑assembled monolayers (SAMs) bearing either hydrophobic –CH₃ or hydrophilic –OH head groups. While both surfaces exhibit similar average water density, the –CH₃ surface shows a pronounced tail in P v(N) at low N, indicating rare but strongly enhanced low‑density fluctuations. When a modest unfavorable linear potential φ (≈0.5 kBT) is applied, the –CH₃ surface’s P v(N) collapses toward N ≈ 0, effectively drying the interface, whereas the –OH surface responds only with a gradual reduction in the mean water number. The susceptibility ∂⟨N⟩/∂φ peaks sharply for the hydrophobic surface, a hallmark of a nanoscopic phase transition.
Having validated the methodology, the authors turn to biologically relevant, irregularly shaped hydrophobic patches. They examine a subunit of the enzyme BphC, which contains a large hydrophobic patch at the domain‑domain interface. In volumes placed near the hydrophilic region, P v(N) is Gaussian and bulk‑like; near the hydrophobic patch, low‑N fluctuations are strongly enhanced. When the protein’s partial charges are turned off—an artificial perturbation that removes favorable electrostatic water‑protein interactions—the low‑N tail becomes even more pronounced, indicating an increase in effective hydrophobicity.
Next, the authors probe the inter‑domain space by translating one domain toward the other at separations of 0.8, 0.6, and 0.4 nm. Even at the smallest gap, the average water count remains high, suggesting the region stays wet. However, all three separations display enhanced low‑N probability, meaning the water is still close to a dewetting transition on the wet side. When electrostatic interactions are disabled, the same 0.6 nm gap exhibits a P v(N) peaked at N ≈ 0, i.e., the water has dried. This demonstrates that a small perturbation can tip the system across the transition.
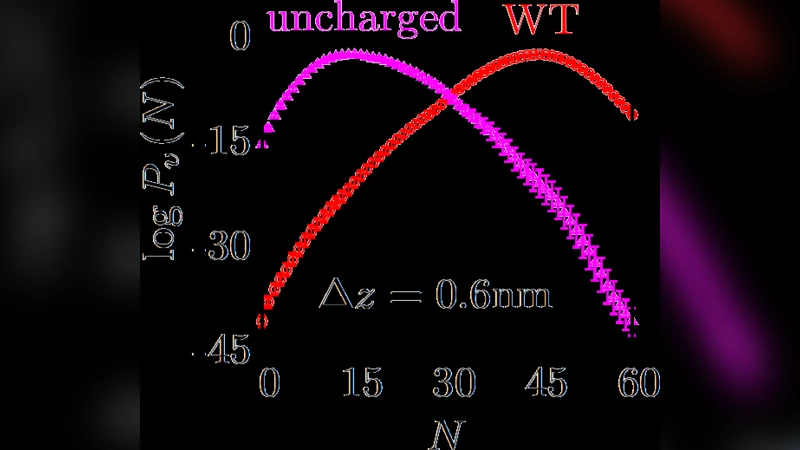
The paper then studies melittin dimers, a system previously reported to sit on the dry side of the transition. For wild‑type dimers, decreasing the separation below ~0.7 nm produces a clear dewetting transition, with P v(N) becoming bimodal (wet and dry basins) at intermediate distances, indicating a free‑energy barrier between the two states. A single point mutation (Ile20→Gly) subtly alters the hydrophobic character of the interface; despite almost identical P v(N) for the isolated dimers, the mutant fails to dewet even at the smallest separation, remaining wet throughout. This highlights how tiny chemical changes can shift the balance of the transition.
Collectively, these results reveal that water density fluctuations—rather than mean density—serve as a sensitive probe of proximity to a dewetting transition. Biomolecules appear to exploit this proximity to regulate binding affinities, assembly kinetics, and functional conformational changes. The authors discuss several broader implications: (i) hydrophobically driven protein‑protein association can be tuned by modest conformational adjustments that alter local water fluctuations; (ii) ion channels such as the mechanosensitive MscS may employ a “vapor‑lock” mechanism where a hydrophobic gate switches between wet (conducting) and dry (non‑conducting) states via small structural rearrangements; (iii) nanofluidic devices and hydrophobic zeolites could harness the same physics for switchable transport or catalytic activity.
In conclusion, the paper provides compelling computational evidence that water adjacent to hydrophobic biomolecular surfaces resides at the brink of a dewetting transition. This positioning endows biological systems with a powerful lever: by modestly perturbing surface topology, chemistry, or environmental conditions, they can dramatically reshape water’s local thermodynamic landscape, thereby modulating intermolecular interactions and functional outcomes. The work bridges a long‑standing gap in our understanding of why some protein surfaces dewet while others do not, and opens avenues for rational design of biomimetic materials and therapeutics that target water‑mediated interactions.
Comments & Academic Discussion
Loading comments...
Leave a Comment