Evaluation of Spatial Resolution and Noise Sensitivity of sLORETA Method for EEG Source Localization Using Low-Density Headsets
Electroencephalography (EEG) has enjoyed considerable attention over the past century and has been applied for diagnosis of epilepsy, stroke, traumatic brain injury and other disorders where 3D localization of electrical activity in the brain is potentially of great diagnostic value. In this study we evaluate the precision and accuracy of spatial localization of electrical activity in the brain delivered by a popular reconstruction technique sLORETA applied to EEG data collected by two commonly used low-density headsets with 14 and 19 measurement channels, respectively. Numerical experiments were performed for a realistic head model obtained by segmentation of MRI images. The EEG source localization study was conducted with a simulated single active dipole, as well as with two spatially separated simultaneously active dipoles, as a function of dipole positions across the neocortex, with several different noise levels in the EEG signals registered on the scalp. The results indicate that while the reconstruction accuracy and precision of the sLORETA method are consistently high in the case of a single active dipole, even with the low-resolution EEG configurations considered in the present study, successful localization is much more problematic in the case of two simultaneously active dipoles. The quantitative analysis of the width of the reconstructed distributions of the electrical activity allows us to specify the lower bound for the spatial resolution of the sLORETA-based 3D source localization in the considered cases.
💡 Research Summary
Electroencephalography (EEG) remains a cornerstone of non‑invasive brain monitoring because of its millisecond temporal resolution and relatively low cost. However, the spatial resolution of EEG is fundamentally limited by the number and distribution of scalp electrodes. The standardized low‑resolution electromagnetic tomography (sLORETA) algorithm is widely used to estimate three‑dimensional current density distributions from scalp potentials, and it is often applied even when only a modest number of channels are available. The present study systematically evaluates how well sLORETA can localize neural sources when the data are recorded with two commercially popular low‑density headsets containing 14 and 19 electrodes, respectively.
A realistic head model was constructed from high‑resolution magnetic resonance images (MRI). The model comprised three tissue compartments—brain gray matter, skull, and scalp—each assigned literature‑based conductivity values. Electrode positions followed a modified 10‑20 system that matches the geometry of the two headsets. Forward simulations were performed using the finite‑element method (FEM) to generate scalp potentials for a set of dipolar sources placed across the neocortical surface. Two experimental conditions were considered: (1) a single active dipole and (2) two simultaneously active dipoles separated by 20 mm, 30 mm, or 40 mm. To mimic realistic recording environments, additive white Gaussian noise was introduced at three signal‑to‑noise ratios (SNR): 20 dB, 10 dB, and 0 dB.
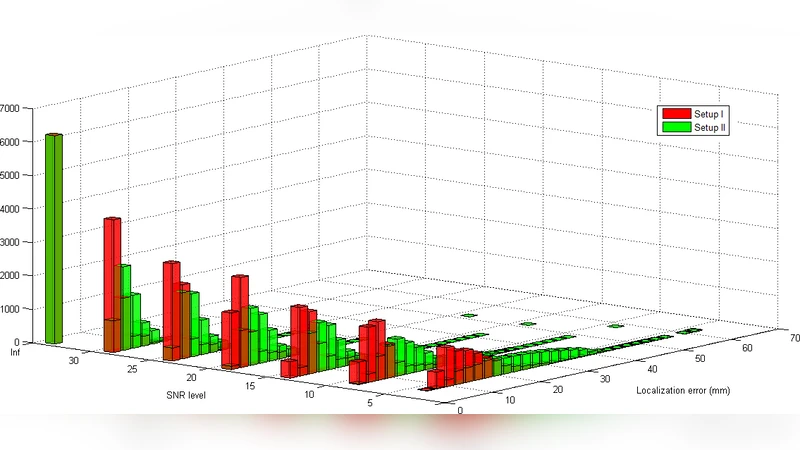
For each simulated dataset, sLORETA was applied to reconstruct the three‑dimensional current density distribution. The primary performance metrics were (a) the Euclidean distance between the true dipole location and the voxel of maximum sLORETA intensity (localization error), (b) the full width at half maximum (FWHM) of the reconstructed distribution (a proxy for spatial spread), and (c) for the two‑dipole condition, the proportion of simulations in which two distinct peaks could be identified.
Results for the single‑dipole scenario showed that both headset configurations achieved high localization accuracy when the SNR was 10 dB or higher. With the 19‑channel headset the mean error was about 5 mm and the average FWHM was 28 mm; the 14‑channel headset yielded slightly larger errors (≈7 mm) and broader spreads (≈32 mm). Even at the harshest noise level (0 dB), the errors increased but remained below 12 mm, indicating that sLORETA is robust to moderate noise when only one source is active.
In contrast, the two‑dipole condition exposed a clear limitation of low‑density recordings. When the dipoles were 40 mm apart, the 19‑channel headset succeeded in separating the two peaks in roughly 70 % of trials at 20 dB SNR, but this success rate fell to below 30 % at 30 mm separation and was essentially zero at 20 mm. The 14‑channel headset performed worse across all separations, achieving peak separation in less than 40 % of trials even at the most favorable SNR. As noise increased, the reconstructed activity merged into a single, broader blob whose FWHM frequently exceeded 45 mm. The average localization error for the two‑dipole case ranged from 15 mm to 25 mm, well beyond the precision required for many clinical applications.
The authors interpret these findings as evidence that sLORETA’s strength lies in providing unbiased, low‑variance estimates for isolated sources, but its spatial resolution is fundamentally constrained by electrode density when multiple sources coexist. The measured lower bound of about 30 mm for the width of sLORETA reconstructions suggests that, with 14–19 channels, the method cannot reliably resolve sources that are closer than roughly one to two centimeters. The study also acknowledges several limitations: the forward model assumes homogeneous conductivity within each tissue class, electrode placement errors and individual head‑shape variability were not modeled, and only white Gaussian noise was considered.
Future work is proposed to explore hybrid approaches that combine sLORETA with beamforming, Bayesian priors, or deep‑learning‑based denoising, as well as to assess the benefits of modestly increasing channel count (e.g., 32‑channel systems) or integrating EEG with structural MRI or functional imaging modalities.
In conclusion, the evaluation demonstrates that low‑density EEG headsets can support accurate single‑source localization with sLORETA, but they fall short when the brain exhibits simultaneous, spatially distinct activations. Clinicians and researchers should therefore be cautious when interpreting multi‑source sLORETA results from 14‑ or 19‑channel recordings and consider augmenting the electrode array or employing complementary imaging techniques to achieve the spatial specificity required for diagnostic or neuroscientific investigations.
Comments & Academic Discussion
Loading comments...
Leave a Comment