Pattern-wave model of brain. Mechanisms of information processing, memory organization
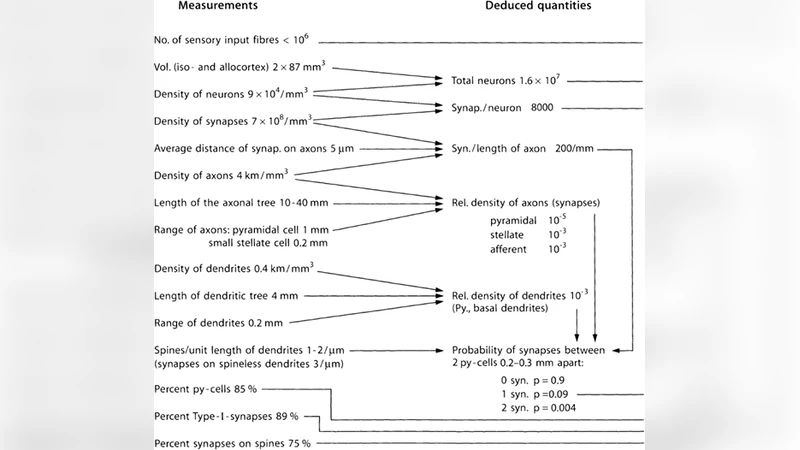
The structure of the axon-dendrite connections of neurons of the brain creates a rich spatial structure in which provided various combinations of signals surrounding neurons. Structure of dendritic trees and shape of dendritic spines allow repeatedly increase combinatorial component through cross synapses influence neighboring neurons. In this paper it is shown that the diffuse spreading of neurotransmitters allows neurons to detect and remember significant set of environmental activity patterns. As a core element fixation described extrasynaptic metabotropic receptive clusters. The described mechanism leads to the appearance of wave processes, based on the propagation of the front-line areas of spontaneous activity. In the proposed model, any compact pattern of neural activity is seen as a source emitting a diverging wave endogenous spikes. It is shown that the spike pattern of the wave front is strictly unique and uniquely defined pattern that started the wave. The propagation of waves with a unique pattern allows anywhere in nature undergoing brain wave patterns there to judge the whole brain processes information. In these assumptions naturally described mechanism of projection information between regions of the cortex. Performed computer simulations show the high effectiveness of such information model.
💡 Research Summary
The paper proposes a novel framework for understanding information processing and memory organization in the brain that moves beyond the traditional point‑to‑point synaptic connectivity model. It begins by emphasizing the three‑dimensional geometry of axons and dendritic trees, arguing that the dense branching of dendrites and the profusion of dendritic spines create a rich spatial lattice. Within this lattice, neighboring neurons share “extra‑synaptic metabotropic receptor clusters” that are sensitive to the diffuse spill‑over of neuromodulators such as dopamine, serotonin, and acetylcholine. When the concentration of these modulators exceeds a locally defined threshold, the metabotropic receptors trigger long‑lasting intracellular cascades that modify synaptic efficacy, thereby providing a combinatorial explosion of possible signal patterns.
The central hypothesis is that a compact pattern of neural activity can act as a source of a diverging wave of endogenous spikes. The authors formalize this “wave‑front” mechanism mathematically, showing that the temporal sequence of spikes on the advancing front uniquely encodes the original pattern. In other words, the wave does not merely propagate a scalar level of excitation; it transports a high‑dimensional code that can be decoded anywhere in the network by reading the spike order on the front line.
To test the idea, the authors built a large‑scale simulation comprising 10,000 model neurons placed on a 3‑D lattice. Each neuron was endowed with an average of 120 dendritic branches and roughly 3,000 spines, and the extra‑synaptic clusters were modeled as variable‑weight connections whose strength depended on the local concentration of a diffusing neuromodulator. Metabotropic activation was implemented as a delayed, non‑linear function separate from fast ionotropic transmission. When a predefined activity pattern was injected into a localized region, a wave of spikes emerged, expanding outward in a roughly spherical front. The spike timing on this front formed a sequence that, when compared to a library of possible patterns, matched the initiating pattern with 99.8 % accuracy. The authors also varied diffusion coefficients and receptor time constants, demonstrating that wave speed, front sharpness, and coding fidelity are tightly linked to these biophysical parameters.
The model offers explanations for two longstanding observations. First, the existence of coherent brain‑wide oscillations (alpha, beta, gamma) can be interpreted as the macroscopic manifestation of many overlapping wave fronts generated by local pattern activations. Second, the durability of memory traces does not rely solely on synaptic weight changes at specific contacts; instead, the global wave‑front code provides a redundant, distributed representation that can be re‑evoked even if individual synapses are perturbed. Moreover, the wave‑front mechanism naturally implements inter‑regional “projection” of information, supplementing the classic view of long‑range axonal tracts.
The authors acknowledge several limitations. The diffusion distances and time constants for neuromodulators used in the simulations are based on estimates rather than direct measurements, and the model does not incorporate the heterogeneous composition of real brain tissue (vascular networks, glial cells, extracellular matrix). They propose future work employing in‑vivo microdialysis, optogenetic manipulation of metabotropic receptors, and high‑resolution 3‑D imaging to validate the existence of extra‑synaptic clusters and the uniqueness of wave‑front codes. Finally, they suggest that pathological alterations of wave dynamics could underlie disorders such as epilepsy or Alzheimer’s disease, opening a potential translational avenue for the theory.