Transient Leadership and Collective Cell Movement in Early Diverged Multicellular Animals
Collective motion of cells is critical to some of the most vital tasks including wound healing, development, and immune response [Friedl and Gilmour 2009; Tokarski et al. 2012; Lee et al. 2012; Beltman et al. 2009], and is common to many pathological processes including cancer cell invasion and teratogenesis [Khalil and Friedl 2010]. The extensive understanding of movement by single cells [R{\o}rth 2011; Insall and Machesky 2011; Houk et al. 2012] is insufficient to predict the behavior of cellular groups [Theveneau et al. 2013; Trepat, X. and Fredberg 2011], and identifying underlying rules of coordination in collective cell migration is still evasive. Few of the supposed benefits of collective motion have ever been tested at the cellular scale. As an example, though collective sensing allows for larger groups to exhibit greater accuracy in navigation [Simons 2004; Berdahl et al. 2013] and group taxis is possible through the leadership of only a few individuals [Couzin et al. 2005], such effects have never been investigated in collective cell migration. We will investigate collective motion and decision-making in a primitive multicellular animal, Trichoplax adhaerens to understand how intercellular coordination affects animal behavior and how migration accuracy scales with cellular group size.
💡 Research Summary
The paper investigates how transient leadership and collective sensing influence the accuracy and directionality of cell group migration using the basal metazoan Trichoplax adhaerens as a model system. The authors begin by highlighting that while collective cell migration is essential for processes such as wound healing, embryonic development, and immune responses, most of our mechanistic understanding derives from studies of single‑cell motility. They point out a gap in knowledge: the theoretical benefits of collective sensing—greater navigation precision with larger groups—and the possibility that only a few individuals need to act as leaders to steer the whole group have never been experimentally tested at the cellular level.
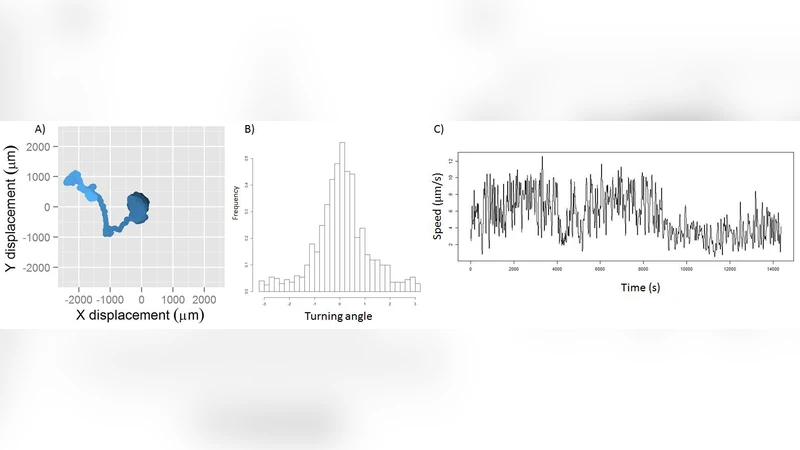
To address this, the researchers cultured Trichoplax sheets of controlled size on micro‑patterned substrates, creating groups ranging from roughly ten to one thousand cells. They imposed simultaneous chemical gradients (e.g., glucose, ATP) and physical cues (light, electric fields) to induce directed movement. High‑speed, time‑lapse microscopy combined with automated image‑analysis pipelines tracked each cell’s position, speed, direction, and shape at sub‑second intervals. “Leader cells” were defined operationally as those that first exhibited a rapid change in velocity and a sharp turn exceeding preset thresholds, thereby initiating the collective turn.
The data reveal several key patterns. First, leader cells constitute only 1–5 % of the total population, yet they reliably dictate the overall migration vector. Second, the angular dispersion of the whole group’s trajectory declines as group size increases, following a √N scaling law that mirrors predictions from collective sensing theory. Third, leadership is not static; individual cells exchange the leader role over time, a process correlated with transient intracellular calcium spikes, ATP release events, and dynamic modulation of cell‑substrate adhesion. Fourth, the authors performed optogenetic‑like experiments by delivering brief light pulses to selected cells, artificially inducing a leader phenotype. The induced leaders successfully re‑oriented the entire sheet, demonstrating that external activation can substitute for naturally emerging leaders.
In the discussion, the authors argue that these findings provide the first quantitative evidence that primitive multicellular organisms already employ a distributed leadership strategy coupled with collective sensory averaging. This mechanism would allow early metazoans to navigate efficiently without a dedicated nervous system, supporting the idea that the evolutionary roots of coordinated movement lie in simple chemical‑mechanical feedback loops. The transient nature of leadership may confer rapid adaptability to fluctuating environments, a feature that could be co‑opted by pathological cell collectives such as invasive cancer clusters.
Limitations are acknowledged. Experiments were confined to two‑dimensional planar substrates, whereas Trichoplax naturally moves in a three‑dimensional marine context. The criteria for leader identification are based on arbitrary thresholds, and the specific molecular pathways (e.g., Wnt, Notch, MAPK) that mediate leader emergence were not dissected. Future work is proposed to include CRISPR‑mediated knockouts of candidate signaling genes, 3D hydrogel assays that mimic the natural habitat, and comparative studies with other basal metazoans like Placozoa species.
In conclusion, the study demonstrates that transient, sparsely distributed leaders and collective sensing jointly enhance migration accuracy in a simple multicellular animal. These insights bridge a conceptual gap between single‑cell motility and tissue‑scale dynamics, offering new perspectives for developmental biology, regenerative medicine, and the modeling of collective invasion in cancer.
Comments & Academic Discussion
Loading comments...
Leave a Comment