Bone refilling in cortical bone multicellular units: Insights into tetracycline double labelling from a computational model
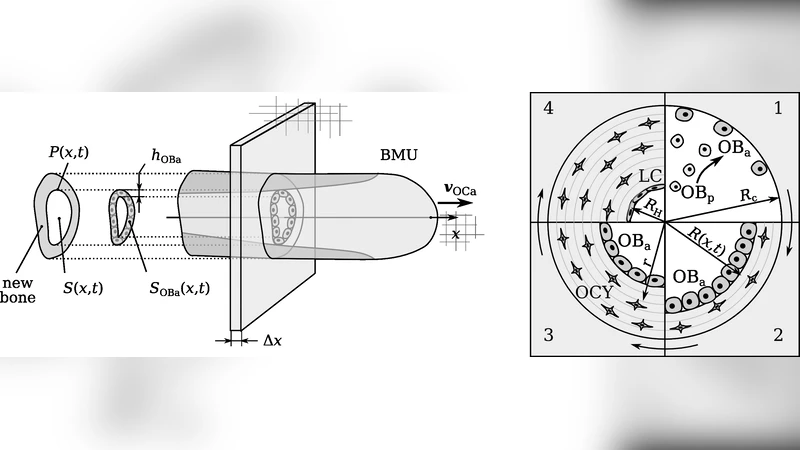
Bone remodelling is carried out by `bone multicellular units’ (BMUs) in which active osteoclasts and active osteoblasts are spatially and temporally coupled. The refilling of new bone by osteoblasts towards the back of the BMU occurs at a rate that depends both on the number of osteoblasts and on their secretory activity. In cortical bone, a linear phenomenological relationship between matrix apposition rate (MAR) and BMU cavity radius is found experimentally. How this relationship emerges from the combination of complex, nonlinear regulations of osteoblast number and secretory activity is unknown. Here, we extend our previous mathematical model of cell development within a single BMU to investigate how osteoblast number and osteoblast secretory activity vary along the BMU’s closing cone. MARs predicted by the model are compared with data from tetracycline double labelling experiments. We find that the linear phenomenological relationship observed in these experiments between MAR and BMU cavity radius holds for most of the refilling phase simulated by our model, but not near the start and end of refilling. This suggests that at a particular bone site undergoing remodelling, bone formation starts and ends rapidly. Our model also suggests that part of the observed cross-sectional variability in tetracycline data may be due to different bone sites being refilled by BMUs at different stages of their lifetime. The different stages of a BMU’s lifetime depend on whether the cell populations within the BMU are still developing or have reached a quasi-steady state while travelling through bone. We find that due to their longer lifespan, active osteoblasts reach a quasi-steady distribution more slowly than active osteoclasts. We suggest that this fact may locally enlarge the Haversian canal diameter (due to a local lack of osteoblasts compared to osteoclasts) near the BMU’s point of origin.
💡 Research Summary
This paper presents a comprehensive mathematical investigation of bone refilling within cortical bone multicellular units (BMUs), focusing on the relationship between matrix apposition rate (MAR) and BMU cavity radius observed in tetracycline double‑labeling experiments. Building on a previously developed single‑BMU model, the authors incorporate the coupled development of active osteoclasts and active osteoblasts, explicitly modeling osteoblast number (Nob) and per‑cell secretory activity (Sob) as functions of spatial position along the BMU’s closing cone and time. Both variables are regulated by nonlinear feedbacks involving growth factors, mechanical stimuli, and cross‑talk between the two cell types. The model discretizes the BMU’s conical geometry into radial coordinates and defines MAR(r,t)=Nob(r,t)·Sob(r,t). Parameter values are calibrated against literature and experimental data, particularly the MAR‑radius pairs obtained from tetracycline double‑labeling.
Simulation outcomes reveal three distinct phases of the refilling process. In the early phase, osteoblast secretory activity rises sharply while cell numbers are still low, producing a pronounced, nonlinear increase in MAR that matches the rapid MAR rise seen in early labeling data. During the middle phase, both osteoclasts and osteoblasts reach a quasi‑steady state; Nob and Sob become approximately constant, and MAR varies linearly with cavity radius (MAR≈α·R+β). This reproduces the experimentally reported linear phenomenological relationship for the majority of the refilling period. In the late phase, osteoblast apoptosis accelerates, Sob declines, and MAR again deviates from linearity, accounting for the observed drop in MAR near the end of refilling.
A notable mechanistic insight is that active osteoblasts, owing to their longer lifespan and slower attainment of steady‑state distribution, lag behind osteoclasts. Consequently, near the BMU’s point of origin, the relative paucity of osteoblasts compared with osteoclasts can transiently enlarge the Haversian canal diameter, providing a quantitative explanation for localized canal widening. The model also suggests that the cross‑sectional variability in tetracycline labeling across different bone sites can be attributed to BMUs being captured at different stages of their lifecycle, rather than intrinsic heterogeneity of the tissue itself.
Overall, the study bridges the gap between complex, nonlinear cellular regulation and the simple linear MAR‑radius relationship observed experimentally. It offers a robust computational framework for interpreting tetracycline labeling data, underscores the importance of accounting for BMU lifecycle stage in clinical and experimental analyses, and proposes testable predictions about canal geometry changes that could inform future histomorphometric investigations and therapeutic strategies targeting bone remodeling.
Comments & Academic Discussion
Loading comments...
Leave a Comment