Endocortical bone loss in osteoporosis: The role of bone surface availability
Age-related bone loss and postmenopausal osteoporosis are disorders of bone remodelling, in which less bone is reformed than resorbed. Yet, this dysregulation of bone remodelling does not occur equally in all bone regions. Loss of bone is more pronounced near and at the endocortex, leading to cortical wall thinning and medullary cavity expansion, a process sometimes referred to as “trabecularisation” or “cancellisation”. Cortical wall thinning is of primary concern in osteoporosis due to the strong deterioration of bone mechanical properties that it is associated with. In this paper, we examine the possibility that the non-uniformity of microscopic bone surface availability could explain the non-uniformity of bone loss in osteoporosis. We use a computational model of bone remodelling in which microscopic bone surface availability influences bone turnover rate and simulate the evolution of the bone volume fraction profile across the midshaft of a long bone. We find that bone loss is accelerated near the endocortical wall where the specific surface is highest. Over time, this leads to a substantial reduction of cortical wall thickness from the endosteum. The associated expansion of the medullary cavity can be made to match experimentally observed cross-sectional data from the Melbourne Femur Collection. Finally, we calculate the redistribution of the mechanical stresses in this evolving bone structure and show that mechanical load becomes critically transferred to the periosteal cortical bone.
💡 Research Summary
The paper addresses why bone loss in osteoporosis and age‑related bone loss is not uniform across the cortical wall but is markedly accentuated at the endocortical surface. The authors hypothesize that the spatial heterogeneity of microscopic bone surface availability (specific surface, sV) drives this non‑uniform remodeling. They construct a one‑dimensional computational model of bone remodeling along the transverse mid‑shaft of a long bone. In the model, the turnover rates for resorption (R) and formation (F) are assumed to be proportional to the local specific surface; thus, regions with higher sV experience faster remodeling. To mimic the systemic shift toward net bone loss observed in aging and post‑menopausal women, the baseline R/F ratio is set slightly above unity (≈1.2), creating a modest bias toward resorption.
The governing equation for the bone volume fraction φ(x,t) incorporates the spatially varying turnover rates and is solved numerically over time. Initial conditions assume a relatively uniform φ across the cortical thickness. As the simulation proceeds, the endocortical region—where sV is highest—shows a rapid decline in φ, while the periosteal side, with lower sV, retains its thickness or even experiences a slight increase. This differential loss leads to thinning of the endocortical wall, expansion of the medullary cavity, and an overall “trabecularisation” of the cortex. By calibrating model parameters, the predicted changes in cortical thickness and medullary area closely match cross‑sectional data from the Melbourne Femur Collection, providing empirical validation of the hypothesis.
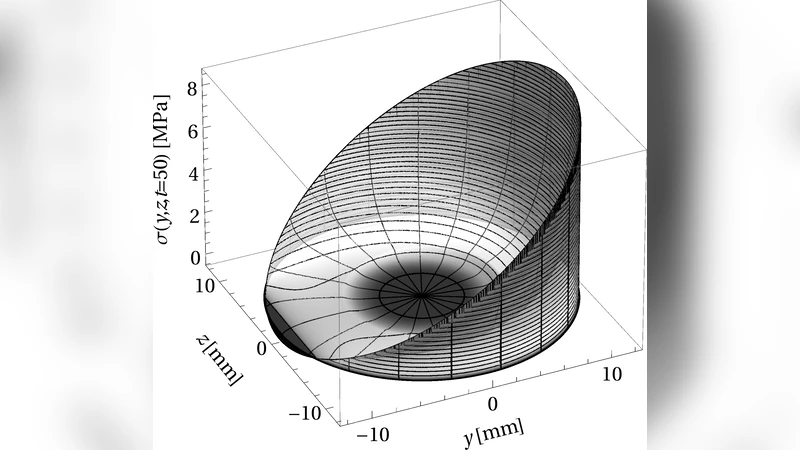
Mechanical consequences are examined using linear elastic finite‑element analysis on the evolving cross‑section. As the endocortical wall thins, its contribution to load bearing diminishes, causing a redistribution of stresses toward the periosteal cortex. The periosteal region consequently experiences higher von Mises stresses, indicating a shift in mechanical function that may predispose this outer layer to microdamage and fracture under normal physiological loads.
The study’s strengths lie in its explicit linkage of microscopic surface geometry to macroscopic remodeling dynamics and its quantitative agreement with experimental morphometric data. However, limitations are acknowledged: the 1‑D representation cannot capture the full three‑dimensional anisotropic architecture of cortical bone; the assumed linear relationship between sV and cellular activity may oversimplify complex biochemical signaling; and systemic factors such as hormonal changes, nutrition, and mechanical loading history are not incorporated.
Future work is suggested to extend the model to full 3‑D finite‑element frameworks, to experimentally determine the non‑linear sV‑cell response, and to integrate additional physiological modulators. Moreover, therapeutic implications are discussed: interventions that reduce endocortical surface exposure (e.g., anti‑resorptive drugs, targeted mechanical loading) or that reinforce the periosteal cortex (e.g., resistance training, anabolic agents) could mitigate the deleterious stress redistribution and improve fracture resistance in osteoporotic patients.