The influence of bone surface availability in bone remodelling - A mathematical model including coupled geometrical and biomechanical regulations of bone cells
Bone is a biomaterial undergoing continuous renewal. The renewal process is known as bone remodelling and is operated by bone-resorbing cells (osteoclasts) and bone-forming cells (osteoblasts). Both biochemical and biomechanical regulatory mechanisms have been identified in the interaction between osteoclasts and osteoblasts. Here we focus on an additional and poorly understood potential regulatory mechanism of bone cells, that involves the morphology of the microstructure of bone. Bone cells can only remove and replace bone at a bone surface. However, the microscopic availability of bone surface depends in turn on the ever-changing bone microstructure. The importance of this geometrical dependence is unknown and difficult to quantify experimentally. Therefore, we develop a sophisticated mathematical model of bone cell interactions that takes into account biochemical, biomechanical and geometrical regulations. We then investigate numerically the influence of bone surface availability in bone remodelling within a representative bone tissue sample. The interdependence between the bone cells’ activity, which modifies the bone microstructure, and changes in the microscopic bone surface availability, which in turn influences bone cell development and activity, is implemented using a remarkable experimental relationship between bone specific surface and bone porosity. Our model suggests that geometrical regulation of the activation of new remodelling events could have a significant effect on bone porosity and bone stiffness. On the other hand, geometrical regulation of late stages of osteoblast and osteoclast differentiation seems less significant. We conclude that the development of osteoporosis is probably accelerated by this geometrical regulation in cortical bone, but probably slowed down in trabecular bone.
💡 Research Summary
Bone is a dynamic tissue that continuously renews itself through the coordinated actions of osteoclasts (bone‑resorbing cells) and osteoblasts (bone‑forming cells). While the biochemical pathways (RANK‑L/OPG, TGF‑β, M‑CSF, etc.) and the biomechanical feedback (strain energy density, fluid flow) that regulate these cells are well documented, the role of the bone’s micro‑geometrical environment—specifically the amount of bone surface that is actually available for cellular activity—has remained largely unexplored. This paper addresses that gap by constructing a comprehensive mathematical model that simultaneously incorporates biochemical, biomechanical, and geometrical regulations of bone cell populations.
The cornerstone of the geometrical component is an experimentally derived relationship between bone porosity (f) and specific surface (S_V, the surface area per unit tissue volume). Empirical data show that S_V follows a power‑law dependence on porosity, S_V = α f^β, where α and β are tissue‑specific constants (different for cortical and trabecular bone). As porosity increases, the available surface expands non‑linearly, providing more “real estate” for osteoclasts and osteoblasts to attach, resorb, and lay down new matrix.
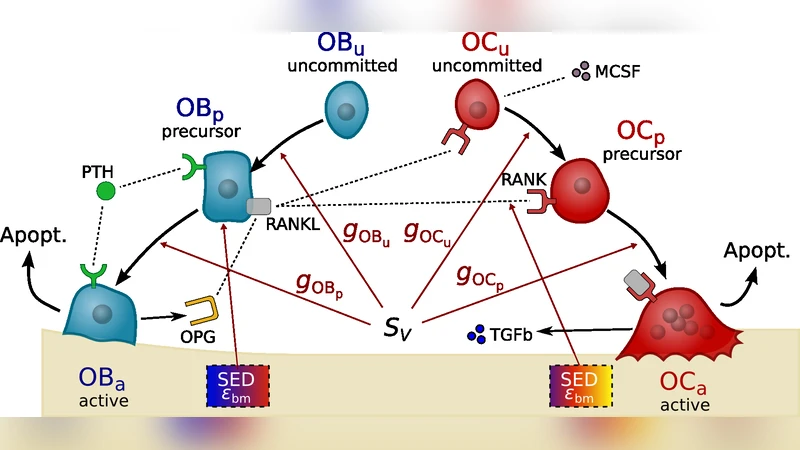
The model consists of a system of ordinary differential equations describing the dynamics of four principal cell compartments: pre‑osteoclasts (P_OC), active osteoclasts (OC), pre‑osteoblasts (P_OB), and active osteoblasts (OB). Transition rates between compartments are modulated by three multiplicative factors:
- Geometrical factor (G_surf) – a function of S_V that scales the activation frequency of new remodeling events (BMU initiation).
- Biomechanical factor (G_mech) – a function of the local strain energy density (ε) sensed by osteocytes, influencing both resorption and formation.
- Biochemical factor (G_bio) – a function of signaling molecules such as RANK‑L/OPG ratio, TGF‑β, and M‑CSF.
For example, the activation rate of pre‑osteoclasts to active osteoclasts is expressed as:
λ_OC = λ_0 · (1 + k_1 S_V) · (1 + k_2 ε) · (1 + k_3 RANKL/OPG)
where λ_0 is a baseline rate and k_1‑k_3 are sensitivity coefficients calibrated from literature. In contrast, the later stages of differentiation (e.g., osteoblast maturation) are assigned a much smaller k_1, reflecting the authors’ hypothesis that surface availability primarily influences the initiation of remodeling rather than the subsequent maturation of the cells.
Parameter values are drawn from a combination of in‑vitro cell‑culture data, animal studies, and human µCT measurements. The model is applied to a representative volume element (RVE) of 1 mm³ for both cortical and trabecular bone. Initial porosities are set to 5 % for cortical bone (low S_V) and 30 % for trabecular bone (high S_V). Two scenarios are simulated: a “normal” homeostatic condition and a “osteoporotic” condition in which the frequency of BMU activation is increased by 50 % to mimic the elevated remodeling observed in post‑menopausal women.
Key Findings
- Geometrical regulation of BMU initiation dominates – In cortical bone, as porosity rises, S_V increases sharply, leading to a non‑linear rise in BMU activation frequency. This accelerates net bone loss, reduces overall stiffness, and reproduces the characteristic cortical thinning seen in osteoporosis.
- Trabecular bone behaves oppositely – The already high S_V in trabecular bone means that additional surface creation preferentially boosts osteoblast activity relative to osteoclasts. Consequently, the same geometrical feedback that aggravates cortical loss actually mitigates trabecular loss, slowing the progression of osteoporosis in the spongy compartment.
- Late‑stage differentiation is less sensitive to surface – Modifying the geometrical factor for the maturation of osteoclasts and osteoblasts (i.e., increasing k_1 for those stages) produces only marginal changes in overall porosity trajectories. This supports the notion that the initial activation step is the critical control point.
- Biomechanical coupling remains essential – Even with strong geometrical feedback, the strain‑energy‑density term continues to modulate the balance between resorption and formation, ensuring that mechanical loading can partially offset the deleterious effects of increased surface area.
Implications
The model suggests that therapeutic strategies for osteoporosis should be tissue‑specific. In cortical bone, interventions that blunt the sensitivity of BMU initiation to surface availability—such as potent RANKL inhibitors or agents that reduce osteocyte‑mediated signaling of surface expansion—could decelerate disease progression. Conversely, in trabecular bone, preserving or modestly enhancing surface area (e.g., through controlled micro‑damage that stimulates remodeling) might be beneficial, provided that osteoblast activity remains dominant.
Moreover, because the model directly accepts porosity data derived from clinical imaging (µCT, HR‑pQCT), it offers a pathway toward patient‑specific predictions of remodeling trajectories and treatment outcomes. Future work could extend the framework to simulate fracture healing, implant integration, or the effects of pharmacological agents that specifically target the geometrical feedback loop.
In summary, by integrating a rigorously quantified geometrical feedback mechanism with established biochemical and biomechanical controls, this study provides a more complete picture of bone remodeling dynamics. It reveals that the availability of bone surface is a potent regulator of remodeling initiation, accelerating cortical bone loss while potentially protecting trabecular bone, thereby offering new insights into the heterogeneous nature of osteoporosis progression.
Comments & Academic Discussion
Loading comments...
Leave a Comment