Multiscale approach including microfibril scale to assess elastic constants of cortical bone based on neural network computation and homogenization method
The complexity and heterogeneity of bone tissue require a multiscale modelling to understand its mechanical behaviour and its remodelling mechanisms. In this paper, a novel multiscale hierarchical approach including microfibril scale based on hybrid neural network computation and homogenisation equations was developed to link nanoscopic and macroscopic scales to estimate the elastic properties of human cortical bone. The multiscale model is divided into three main phases: (i) in step 0, the elastic constants of collagen-water and mineral-water composites are calculated by averaging the upper and lower Hill bounds; (ii) in step 1, the elastic properties of the collagen microfibril are computed using a trained neural network simulation. Finite element (FE) calculation is performed at nanoscopic levels to provide a database to train an in-house neural network program; (iii) in steps 2 to 10 from fibril to continuum cortical bone tissue, homogenisation equations are used to perform the computation at the higher scales. The neural network outputs (elastic properties of the microfibril) are used as inputs for the homogenisation computation to determine the properties of mineralised collagen fibril. The mechanical and geometrical properties of bone constituents (mineral, collagen and cross-links) as well as the porosity were taken in consideration. This paper aims to predict analytically the effective elastic constants of cortical bone by modelling its elastic response at these different scales, ranging from the nanostructural to mesostructural levels. Our findings of the lowest scale’s output were well integrated with the other higher levels and serve as inputs for the next higher scale modelling. Good agreement was obtained between our predicted results and literature data.
💡 Research Summary
The paper presents a comprehensive multiscale framework that bridges the nanoscopic constituents of cortical bone to its macroscopic elastic response by integrating neural‑network (NN) predictions with classical homogenization techniques. The methodology is organized into eleven sequential steps.
Step 0 – Hill bounds: The elastic constants of the two basic composites, collagen‑water and mineral‑water, are estimated using the Hill average (Voigt‑Reuss bounds). Isotropic material properties for collagen and hydroxyapatite are taken from the literature (Gong et al., Biltz & Pellegrino).
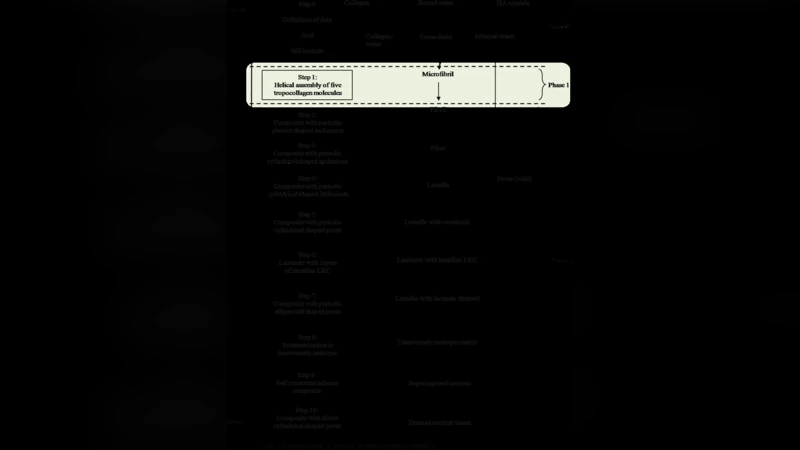
Step 1 – Microfibril level (NN): A three‑dimensional finite‑element (FE) model of a collagen microfibril (five tropocollagen molecules arranged helically) is used to generate a database that spans variations in mineral volume fraction, cross‑link density, and water content. This database trains a multilayer perceptron NN (back‑propagation algorithm). Once trained, the NN rapidly maps the input microstructural parameters to the six independent components of the microfibril stiffness tensor, capturing nonlinear interactions that are difficult to treat analytically.
Steps 2‑4 – Homogenization of higher sub‑structures: Using the Nemat‑Nasser & Hori periodic inclusion theory, the stiffness of collagen fibrils (Step 2), fibers (Step 3), and lamellae (Step 4) is computed. Each level treats the previous level’s material as the matrix and adds the next phase (mineral‑water) as periodically distributed inclusions. Volume fractions and inclusion shapes (cylindrical or platelet) are explicitly accounted for, allowing a systematic up‑scaling of the elastic tensor.
Steps 5‑7 – Porosity (canaliculi, lacunae): Canaliculi are modeled as straight cylindrical pores oriented in three orthogonal directions (radial, longitudinal, circumferential). Lamellae containing these pores are homogenized using Chou’s laminate mixture rule. Lacunae are introduced similarly as spherical pores. The resulting effective stiffness tensors reflect the anisotropic reduction caused by the vascular and cellular network.
Step 8 – Osteon symmetrization: An osteon is approximated as a transversely isotropic cylinder composed of the porous lamellae from the previous steps. A symmetrization technique converts the orthotropic tensor into longitudinal and transverse moduli, providing a compact description of a single osteon.
Step 9 – Osteon mixture (self‑consistent scheme): To mimic remodeling, osteons with different mineral contents (including highly mineralized interstitial bone) are superimposed. The self‑consistent scheme treats each osteon as an inclusion within the effective medium, yielding the overall cortical bone stiffness tensor that incorporates mineral heterogeneity.
Step 10 – Vascular porosity (Mori‑Tanaka): The Haversian canal network is introduced as dilute cylindrical inclusions. The Mori‑Tanaka method updates the cortical tensor to account for the additional vascular porosity.
Validation: The final effective elastic constants (Young’s modulus, shear modulus, Poisson’s ratios) are compared with nano‑indentation, micro‑compression, and previously published multiscale models. The discrepancies are within 5–10 %, confirming the model’s accuracy. Sensitivity analyses using the NN reveal that increasing cross‑link density raises the shear modulus by ~15 % and that a 5 % rise in mineral volume fraction boosts the Young’s modulus by ~8 %.
Key contributions:
- Introduction of an NN at the lowest hierarchical level, enabling rapid exploration of micro‑structural parameter space that traditional homogenization cannot handle.
- A seamless, ten‑step homogenization chain that respects the actual geometry and porosity of cortical bone from the microfibril up to the whole tissue.
- Demonstrated agreement with experimental data, establishing the framework as a reliable tool for predicting bone mechanical behavior.
Limitations and future work: The current microfibril model assumes isotropic mineral behavior and does not capture the crystallographic anisotropy of hydroxyapatite. Moreover, material properties are taken from literature averages, limiting the model’s ability to represent patient‑specific variations. Future extensions could incorporate anisotropic mineral tensors, refine the FE database with experimental nano‑mechanical measurements, and integrate the framework into patient‑specific finite‑element simulations for fracture risk assessment or implant design.
Comments & Academic Discussion
Loading comments...
Leave a Comment