Stable all nitrogen metallic salt at terapascal pressures
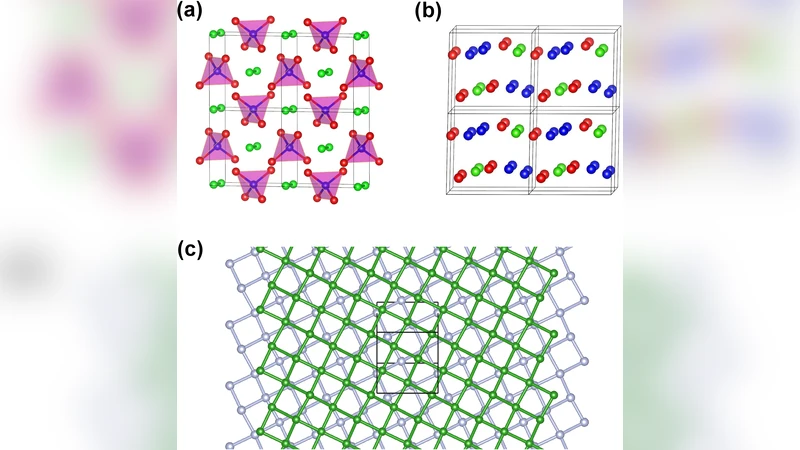
The phase diagram and equation of state of dense nitrogen are of interest in understanding the fundamental physics and chemistry under extreme conditions, including planetary processes, and in discovering new materials. We predict several stable phases of nitrogen at multi-TPa pressures, including a P4/nbm structure consisting of partially charged N2 pairs and N5 tetrahedra, which is stable in the range 2.5-6.8 TPa. This is followed by a modulated layered structure between 6.8 and 12.6 TPa, which also exhibits a significant charge transfer. The P4/nbm metallic nitrogen salt and the modulated structure are stable at high pressures and temperatures, and they exhibit strongly ionic features and charge density distortions, which is unexpected in an element under such extreme conditions and could represent a new class of nitrogen materials. The P-T phase diagram of nitrogen at TPa pressures is investigated using quasiharmonic phonon calculations and ab initio molecular dynamics simulations.
💡 Research Summary
The authors investigate the behavior of elemental nitrogen under extreme compression, reaching pressures in the terapascal (TPa) range, a regime relevant to the interiors of massive planets and to the discovery of novel high‑density materials. Using state‑of‑the‑art crystal‑structure prediction methods (CALYPSO and USPEX) combined with density‑functional theory (DFT) calculations, they identify several previously unknown stable nitrogen phases that display metallic conductivity together with pronounced charge separation—features that are highly unusual for a single element under such conditions.
The first of these phases, stable between 2.5 TPa and 6.8 TPa, adopts a tetragonal P4/nbm symmetry. Its crystal lattice consists of two distinct building blocks: partially charged N₂ dimers and N₅ tetrahedral units. Bader charge analysis reveals that the N₂ pairs carry a modest positive charge (≈ +0.4 e) while the N₅ tetrahedra acquire a negative charge (≈ –0.8 e), indicating a net transfer of electrons from the dimers to the tetrahedra. This internal charge redistribution creates an “ionic” character within a metallic framework, a situation the authors term a “metallic nitrogen salt.” Band‑structure calculations show that the Fermi level intersects dispersive bands, confirming metallic behavior despite the presence of distinct ionic substructures.
When the pressure exceeds 6.8 TPa, the system undergoes a structural transition to a modulated layered phase that remains stable up to about 12.6 TPa. In this phase the nitrogen atoms arrange into alternating layers, and the electron density forms thin, quasi‑two‑dimensional conduction channels between the layers. The charge separation persists, with the layers containing N₂‑derived motifs remaining slightly positive and those built from N₅‑derived motifs remaining negative. This layered arrangement further enhances anisotropic electronic transport while preserving the ionic contrast.
To assess thermodynamic stability, the authors perform quasiharmonic phonon calculations, obtaining free‑energy curves as functions of pressure and temperature. Both the P4/nbm and the layered modulated structures retain dynamical stability up to at least 2000 K, as confirmed by ab‑initio molecular‑dynamics (AIMD) simulations. By combining these results they construct a pressure‑temperature phase diagram that delineates the stability fields of the newly identified phases, the transition boundaries, and the regions where they coexist with previously known high‑pressure nitrogen phases (e.g., ζ‑N₂, ε‑N₂).
The discovery of a metallic, yet strongly ionic, nitrogen phase challenges the conventional view that elemental solids under extreme compression become uniformly metallic and electron‑delocalized. Instead, nitrogen demonstrates that even a single‑element system can develop internal charge ordering, effectively behaving as a self‑assembled salt. This insight has several implications:
-
Planetary Science – In the deep interiors of giant planets where pressures exceed several TPa, nitrogen could contribute to electrical conductivity and affect magnetic field generation. Its ionic character may also influence the solubility of other light elements.
-
Materials Design – The identified structures suggest routes to synthesize ultra‑dense, conductive, and potentially superhard nitrogen‑based materials, exploiting the combination of metallic bonding and strong directional N–N interactions.
-
Fundamental Physics – The work expands the taxonomy of high‑pressure phases, showing that charge transfer and ionic motifs are not exclusive to compounds but can emerge intrinsically in elemental lattices.
Overall, the paper provides a comprehensive computational study that predicts a new class of nitrogen materials—metallic salts formed by the element itself—stable over a broad pressure‑temperature window. The findings open avenues for experimental verification using dynamic compression techniques (laser‑driven shock or ramp compression) and for exploring analogous behavior in other light elements under terapascal conditions.