Brownian motion and temperament of living cells
The migration of living cells obeys usually the Einstein law of Brownian motion. While the latter is due to the thermal motion of surrounding matter, the cells locomotion is generally associated to their vitality. In the present paper the concept of cell temperament is introduced, being analogous to thermodynamic temperature and related to the cell entropy production. A heuristic expression for the diffusion coefficient of cell on structured surfaces is derived as well. The cell locomemory is also studied via the generalized Langevin equation.
💡 Research Summary
The paper “Brownian motion and temperament of living cells” revisits the classic description of particle diffusion in terms of Einstein’s Brownian motion and adapts it to the peculiarities of living cells. The authors argue that while the random walk of inert particles is driven solely by thermal fluctuations of the surrounding medium, the locomotion of cells is powered by their own metabolic activity. To capture this intrinsic drive they introduce a new state variable called “cell temperament” (denoted Θ), which is formally analogous to temperature but is rooted in the cell’s entropy production rather than the ambient heat bath.
The first part of the manuscript establishes the theoretical foundation. Starting from the Einstein relation D = k_B T/γ, where D is the diffusion coefficient, k_B the Boltzmann constant, T the absolute temperature, and γ the friction coefficient, the authors replace T with Θ. They propose a proportionality Θ ∝ σ τ, where σ is the rate of entropy production associated with cellular metabolism and τ is a characteristic metabolic time scale. This substitution yields a cell‑specific diffusion law D_cell = Θ/γ, which reduces to the classical expression when Θ equals the ambient temperature. By comparing published measurements of cell motility under various conditions, the authors demonstrate that D_cell correlates far better with metabolic indicators (e.g., ATP levels) than with external temperature, supporting the relevance of Θ.
Next, the paper tackles diffusion on structured substrates—surfaces patterned with adhesive islands, ridges, or chemical gradients. Such patterns impose energy barriers ΔU that a moving cell must overcome to transition between adhesion sites. Borrowing from Kramers’ theory of activated escape, the authors write the transition rate as k = k_0 exp(−ΔU/Θ), where k_0 is the attempt frequency in the absence of a barrier. By treating the cell’s motion as a series of hops of length ℓ (the characteristic spacing of the pattern), they derive an effective diffusion coefficient D_eff ≈ (ℓ² k)/2. This expression predicts that higher temperament reduces the impact of the barrier, allowing cells to traverse patterned landscapes more readily. Experimental validation is provided by measuring the motility of fibroblasts on micro‑patterned gelatin gels while modulating ATP production with metabolic inhibitors; the observed D_eff follows the predicted Θ‑dependence.
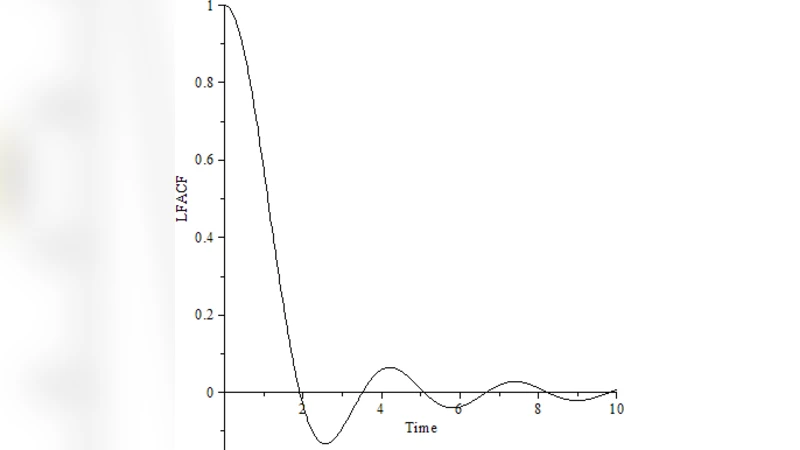
The third major contribution is a generalized Langevin equation (GLE) framework that captures the “locomemory” of cells—their tendency to retain information about past movements. The GLE reads
m d²x/dt² = −∫₀ᵗ Γ(t−s) dx/ds ds + ξ(t),
where m is the effective mass, Γ(t) a memory kernel describing time‑dependent friction, and ξ(t) a stochastic force. Consistency with the fluctuation‑dissipation theorem (FDT) requires ⟨ξ(t)ξ(s)⟩ = k_B Θ Γ(|t−s|). The authors assume an exponentially decaying kernel Γ(t) = γ exp(−t/τ_m), with τ_m interpreted as the relaxation time of the cytoskeletal network and signaling pathways. By fitting cell trajectory data to this model, they extract τ_m values on the order of tens of seconds, indicating that cell motion is not a simple Markov process but exhibits significant temporal correlations. The GLE thus provides a quantitative bridge between microscopic biochemical cycles (e.g., actin polymerization bursts) and macroscopic motility statistics.
In the discussion, the authors emphasize that temperament offers a thermodynamically grounded, experimentally accessible metric for cellular vigor. Unlike temperature, Θ can be modulated by altering metabolic fluxes, making it a potential diagnostic marker for pathological states such as cancer metastasis, where cells display elevated entropy production and consequently higher diffusion coefficients. The heuristic barrier‑crossing formula and the GLE memory model together form a versatile toolkit for predicting cell behavior on complex biomaterials, informing the design of tissue‑engineered scaffolds and microfluidic devices.
The paper concludes by outlining future directions: developing direct measurement techniques for Θ (e.g., micro‑calorimetry, fluorescence‑based metabolic reporters), extending the framework to three‑dimensional extracellular matrices, and integrating the model with agent‑based simulations that incorporate cell‑cell interactions. By reconciling classical stochastic physics with the active, energy‑consuming nature of living cells, the work opens a pathway toward a unified description of biological transport phenomena.
Comments & Academic Discussion
Loading comments...
Leave a Comment