Rhythmogenic neuronal networks, pacemakers, and k-cores
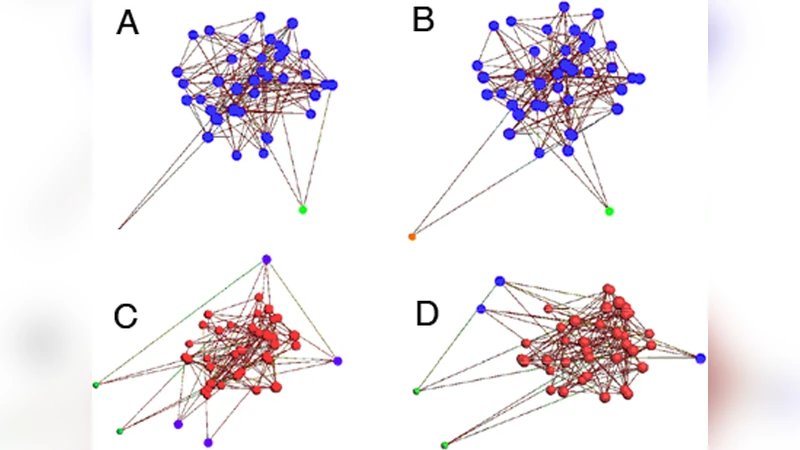
Neuronal networks are controlled by a combination of the dynamics of individual neurons and the connectivity of the network that links them together. We study a minimal model of the preBotzinger complex, a small neuronal network that controls the breathing rhythm of mammals through periodic firing bursts. We show that the properties of a such a randomly connected network of identical excitatory neurons are fundamentally different from those of uniformly connected neuronal networks as described by mean-field theory. We show that (i) the connectivity properties of the networks determines the location of emergent pacemakers that trigger the firing bursts and (ii) that the collective desensitization that terminates the firing bursts is determined again by the network connectivity, through k-core clusters of neurons.
💡 Research Summary
The paper presents a minimalist theoretical and computational study of the pre‑Bötzinger complex (preBötC), a small excitatory neuronal network that generates the rhythmic breathing pattern in mammals. The authors construct a model consisting of N ≈ 200 identical excitatory neurons whose dynamics are reduced to a two‑state (resting ↔ firing) description. Synaptic connections are generated as an Erdős‑Rényi random graph with connection probability p, allowing the authors to systematically vary network density while keeping all neuronal parameters identical. Each neuron fires when its total synaptic input exceeds a fixed threshold; after firing it enters a refractory/desensitized state during which its excitability is reduced in proportion to the cumulative activity of its presynaptic partners. This simple framework captures two essential biological processes: (i) the initiation of a burst by a subset of highly connected neurons, and (ii) the termination of the burst through collective desensitization.
Key Findings
-
Emergent Pacemakers Are Determined by Connectivity
In simulations where p exceeds a modest value, the network exhibits spontaneous, periodic burst episodes. Remarkably, despite the homogeneity of neuronal parameters, a small number of neurons with above‑average degree consistently act as “pacemakers”: they reach threshold first, fire, and then recruit the rest of the network into a synchronized burst. The location of these pacemakers is not pre‑assigned; it emerges from the random wiring. Statistical analysis shows that pacemaker neurons typically have a degree 1.5–2 times larger than the network mean, and their immediate neighborhoods become rapidly synchronized, providing the seed for the global oscillation. -
Burst Termination Is Governed by k‑Core Collapse
As a burst proceeds, each neuron’s desensitization accumulates, lowering its effective input gain. The authors map this process onto the graph‑theoretic concept of a k‑core: the maximal subgraph in which every node has at least k connections within the subgraph. During a burst, the effective functional connectivity of the network shrinks; when the active subnetwork can no longer sustain a k‑core of a critical order (e.g., k = 3 or 4), the collective firing collapses and the burst terminates. Thus, the end of a rhythmic episode is not a simple exhaustion of individual neurons but a structural breakdown of a highly connected core. -
Deviation from Mean‑Field Predictions
Classical mean‑field treatments of excitatory networks predict a single critical connection probability p_c above which the system exhibits sustained activity, and a monotonic relationship between p and burst frequency. The present study demonstrates two crucial departures: (a) bursts can arise even when p < p_c if a few high‑degree nodes exist, and (b) bursts may cease despite p ≫ p_c if the k‑core structure is weak (i.e., the network lacks a robust high‑order core). Consequently, the distribution of degrees—and not merely its average—determines both the onset and offset of rhythmic activity.
Biological Relevance
Experimental work on the preBötC has identified “kernel” or “hub” neurons that, when optogenetically stimulated, can trigger inspiratory bursts, as well as a gradual decline of inspiratory drive during each cycle. The model reproduces these observations: emergent pacemakers correspond to hub neurons, while the k‑core collapse mirrors the observed progressive loss of excitability that ends each inspiratory phase. This alignment suggests that therapeutic interventions (pharmacological agents, electrical or optical stimulation) should target the network topology—specifically, the high‑degree hub population and the integrity of the k‑core—rather than merely modulating overall excitability.
Implications and Future Directions
The work underscores the necessity of incorporating network topology into any dynamical description of small neuronal circuits. It also provides a concrete framework for predicting how alterations in connectivity (e.g., due to development, injury, or disease) might affect breathing rhythmogenesis. Future studies could integrate experimentally measured connectivity matrices from the preBötC, explore heterogeneous neuronal types (e.g., inhibitory interneurons), and examine how neuromodulators reshape the k‑core structure. Ultimately, such refined models may guide the design of closed‑loop ventilatory support systems or targeted neuromodulation therapies for respiratory disorders such as central sleep apnea or congenital hypoventilation syndrome.
Comments & Academic Discussion
Loading comments...
Leave a Comment