Directional interactions and cooperativity between mechanosensitive membrane proteins
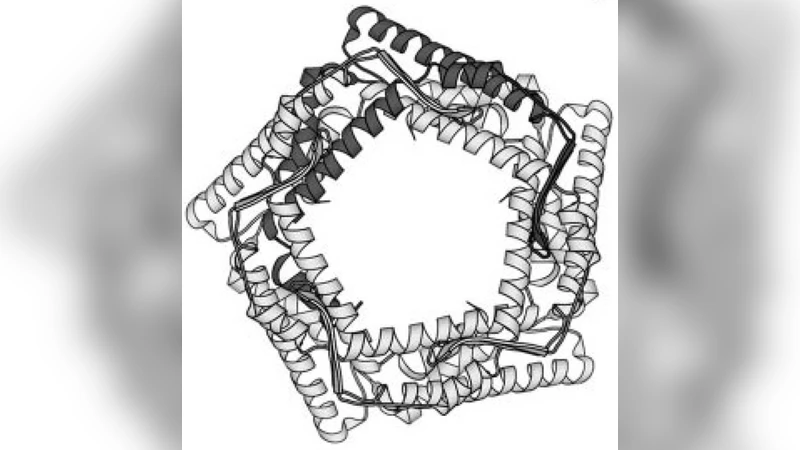
While modern structural biology has provided us with a rich and diverse picture of membrane proteins, the biological function of membrane proteins is often influenced by the mechanical properties of the surrounding lipid bilayer. Here we explore the relation between the shape of membrane proteins and the cooperative function of membrane proteins induced by membrane-mediated elastic interactions. For the experimental model system of mechanosensitive ion channels we find that the sign and strength of elastic interactions depend on the protein shape, yielding distinct cooperative gating curves for distinct protein orientations. Our approach predicts how directional elastic interactions affect the molecular structure, organization, and biological function of proteins in crowded membranes.
💡 Research Summary
The paper investigates how the physical shape of membrane proteins influences their cooperative behavior through membrane‑mediated elastic interactions. Using a continuum elastic model of the lipid bilayer, the authors treat proteins as embedded inclusions with defined curvature and thickness profiles. The interaction energy between two proteins is derived as a function of their center‑to‑center distance (r) and relative orientation (θ). By parameterizing the model with high‑resolution structures of mechanosensitive channels—MscL (largely cylindrical) and MscS (highly asymmetric)—the study quantifies how non‑spherical geometry creates anisotropic deformation fields in the surrounding membrane.
Key findings include:
-
Shape‑dependent interaction sign – Cylindrical proteins generate a simple distance‑dependent attraction or repulsion that scales roughly as 1/r⁴. In contrast, asymmetric proteins produce a mixed potential whose sign flips with rotation angle; when two asymmetric inclusions are aligned (θ≈0°) the interaction is attractive, whereas anti‑aligned configurations (θ≈180°) become repulsive. This arises because the asymmetric shape imposes a directional strain on the bilayer, breaking the isotropy of the deformation field.
-
Cooperative gating curves – When two channels are within ~5–10 nm, the membrane‑mediated interaction modifies the effective gating tension. Aligned channels experience a reduction of the gating threshold by 15–25 % relative to isolated channels, leading to cooperative opening. Conversely, anti‑aligned channels require higher tension, effectively suppressing each other’s opening. The authors translate these effects into a cooperativity coefficient that can be directly compared with electrophysiological measurements.
-
Critical distance and angular window – The cooperative effect decays rapidly beyond ~12 nm and is strongest within a ±30° angular window. This quantitative prediction matches the typical protein density in bacterial membranes, suggesting that such elastic cooperativity is physiologically relevant.
-
Predictive power for mutations and lipid composition – By altering the geometric parameters (e.g., truncating a peripheral helix or changing the hydrophobic mismatch), the model predicts how specific mutations or changes in lipid tail length/composition will shift the interaction landscape. This provides a framework for rational design of synthetic membranes or engineered channels with tailored cooperative properties.
The authors propose experimental validation strategies: (i) Förster resonance energy transfer (FRET) between fluorescently labeled channels to monitor distance‑ and orientation‑dependent proximity, (ii) patch‑clamp recordings on reconstituted bilayers where membrane tension can be precisely controlled while varying channel density, and (iii) high‑resolution atomic force microscopy or cryo‑EM to visualize the spatial arrangement of channels in native‑like membranes.
Biologically, the work suggests that mechanosensitive channels do not act as isolated sensors; instead, they form a mechanically coupled network that can fine‑tune cellular responses to osmotic or mechanical stress. The directional nature of the interaction implies that cells could regulate sensitivity by organizing channels in specific patterns or by modulating lipid composition to alter the elastic constants of the bilayer.
In conclusion, the study provides a rigorous theoretical framework linking protein geometry, membrane elasticity, and functional cooperativity. It demonstrates that anisotropic elastic interactions can generate distinct gating behaviors for different protein orientations, offering a mechanistic explanation for how crowded membranes orchestrate complex biological functions. Future extensions could incorporate nonlinear elasticity, membrane curvature, and multi‑protein ensembles, thereby deepening our understanding of the mechanical dimension of membrane protein biology.
Comments & Academic Discussion
Loading comments...
Leave a Comment