Structure-property-function relationships in triple helical collagen hydrogels
In order to establish defined biomimetic systems, type I collagen was functionalised with 1,3-Phenylenediacetic acid (Ph) as aromatic, bifunctional segment. Following investigation on molecular organization and macroscopic properties, material functionalities, i.e. degradability and bioactivity, were addressed, aiming at elucidating the potential of this collagen system as mineralization template. Functionalised collagen hydrogels demonstrated a preserved triple helix conformation. Decreased swelling ratio and increased thermo-mechanical properties were observed in comparison to state-of-the-art carbodiimide (EDC)-crosslinked collagen controls. Ph-crosslinked samples displayed no optical damage and only a slight mass decrease (~ 4 wt.-%) following 1-week incubation in simulated body fluid (SBF), while nearly 50 wt.-% degradation was observed in EDC-crosslinked collagen. SEM/EDS revealed amorphous mineral deposition, whereby increased calcium phosphate ratio was suggested in hydrogels with increased Ph content. This investigation provides valuable insights for the synthesis of triple helical collagen materials with enhanced macroscopic properties and controlled degradation. In light of these features, this system will be applied for the design of tissue-like scaffolds for mineralized tissue formation.
💡 Research Summary
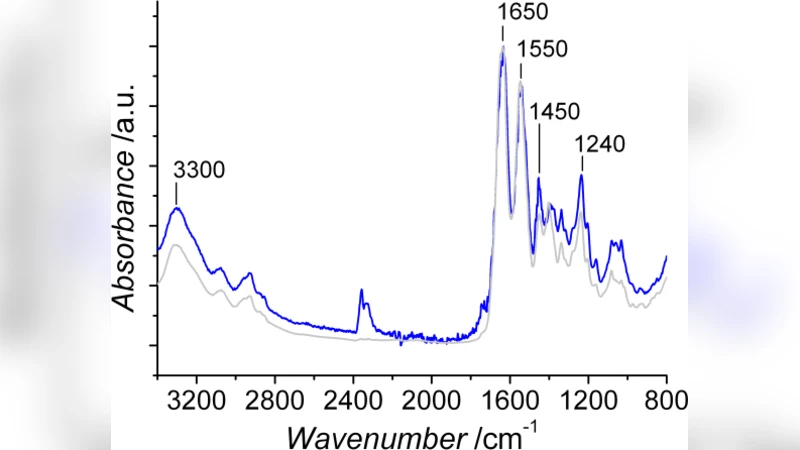
The paper presents a systematic investigation of a novel collagen‑based hydrogel designed to serve as a biomimetic template for mineralized tissue engineering. Type I collagen was chemically functionalized with 1,3‑phenylenediacetic acid (Ph), an aromatic bifunctional linker, and compared to conventional carbodiimide (EDC) cross‑linked collagen hydrogels. The authors first confirmed that the Ph‑cross‑linked network retained the characteristic triple‑helical conformation of native collagen, as demonstrated by FT‑IR, circular dichroism, and Raman spectroscopy. In contrast, EDC cross‑linking induced subtle alterations in the helical signature, suggesting a degree of structural perturbation.
Mechanical and swelling tests revealed that Ph incorporation markedly reduced the equilibrium swelling ratio (from ~250 % in EDC gels to ~120 % in the highest Ph‑content gels) and simultaneously increased the thermal transition temperature (by ~7 °C) and the storage modulus (by up to 80 %). These improvements are attributed to the aromatic, rigid nature of Ph, which creates a denser, more thermally stable network while limiting water uptake.
Degradability was evaluated by immersing the hydrogels in simulated body fluid (SBF) for one week. Ph‑cross‑linked samples exhibited only a modest mass loss (~4 wt %), whereas EDC‑cross‑linked controls degraded by nearly 50 wt %. Optical microscopy showed that Ph gels maintained a smooth, undamaged surface after SBF exposure, indicating superior resistance to hydrolytic attack.
Mineralization potential was assessed by SEM/EDS after SBF incubation. All Ph‑containing hydrogels supported the deposition of an amorphous calcium‑phosphate layer. Importantly, the Ca/P atomic ratio increased with higher Ph content (from ~1.3 to ~1.6), suggesting that the aromatic linker promotes calcium ion binding and facilitates the nucleation of a more bone‑like mineral phase. The mineral coating also contributed to a further increase in mechanical stiffness, highlighting a synergistic effect between the organic scaffold and the inorganic deposit.
Overall, the study demonstrates that Ph‑mediated cross‑linking offers a multifaceted advantage: it preserves the native triple‑helical architecture, enhances thermo‑mechanical performance, dramatically slows enzymatic and hydrolytic degradation, and provides a controllable platform for mineral nucleation. These attributes make Ph‑cross‑linked collagen hydrogels promising candidates for the fabrication of tissue‑engineered scaffolds aimed at bone, dentin, or other mineralized tissue regeneration. Future work is suggested to explore cell‑compatibility, in‑vivo integration, and the incorporation of bioactive ions or growth factors to further tailor the scaffold’s regenerative capacity.
Comments & Academic Discussion
Loading comments...
Leave a Comment