Finite Volume Model to Study the Effect of ER flux on Cytosolic Calcium Distribution in Astrocytes
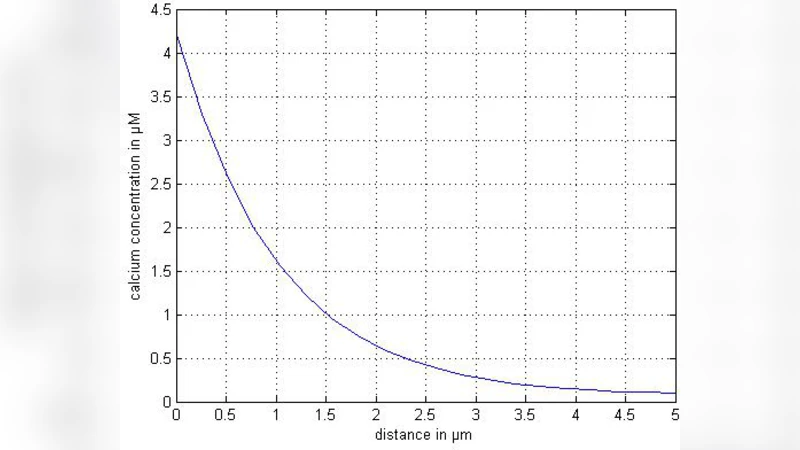
Most of the intra-cellular events involved in the initiation and propagation phases of this process has now been identified astrocytes. The control of the spread of intracellular calcium signaling has been demonstrated to occur at several levels including IP3 receptors, intracellular Ca2+ stores like endoplasmic reticulum (ER) . In normal and pathological situations that affect one or several of these steps can be predicted to influence on astrocytic calcium waves. In view of above a mathematical model is developed to study interdependence of all the important parameters like diffusion coefficient and influx over [Ca2+] profile. Model incorporates the ER fluxes like, leak Pump Chan J J andJ . Finite volume method is employed to solve the problem. A program has been developed using in MATLAB 7.5 for the entire problem and simulated on an AMD-Turion 32-bite machine to compute the numerical results. In view of above a mathematical model is developed to study calcium transport between cytosol and ER.
💡 Research Summary
The paper presents a comprehensive mathematical and numerical investigation of calcium dynamics in astrocytes, focusing on how fluxes between the endoplasmic reticulum (ER) and the cytosol shape the spatial distribution of intracellular Ca²⁺. Recognizing that astrocytic calcium waves are regulated at multiple levels—including IP₃ receptors, ER storage, SERCA pumps, and leak channels—the authors construct a one‑dimensional reaction‑diffusion model that captures the essential physics of calcium exchange. The governing equation for the cytosolic calcium concentration C(x,t) is expressed as
∂C/∂t = D ∂²C/∂x² + J_leak – J_pump + J_chan,
where D denotes the cytosolic diffusion coefficient, J_leak represents passive ER leak, J_pump models active SERCA‑mediated re‑uptake, and J_chan accounts for IP₃‑receptor‑mediated release. Each flux term is formulated with biologically realistic non‑linear kinetics: J_pump follows a Michaelis‑Menten relationship, while J_chan incorporates a Hill function to reflect cooperative IP₃ binding and calcium‑induced calcium release (CICR).
To solve the PDE system, the authors adopt the Finite Volume Method (FVM), which conserves mass across discrete control volumes and is well‑suited for handling fluxes at cell interfaces. The spatial domain is discretized into uniform cells; fluxes at each face are approximated by central differences, and the resulting semi‑discrete equations are integrated in time using an explicit forward‑Euler scheme. Stability is ensured by respecting the Courant–Friedrichs–Lewy (CFL) condition, and Neumann (no‑flux) boundary conditions are imposed at the cell membrane, while a constant external calcium influx is introduced as a source term.
Implementation is carried out in MATLAB 7.5, with a dedicated script that automates parameter sweeps for the diffusion coefficient (D), external influx magnitude (I₀), and the maximal rates of the three ER fluxes (V_max,leak, V_max,pump, V_max,chan). The computational platform—a 32‑bit AMD Turion processor—provides sufficient performance for the extensive simulations required to map the multidimensional parameter space.
Key findings from the numerical experiments are as follows:
- Diffusion Effects – Increasing D smooths the calcium profile, reducing peak amplitudes and accelerating wave propagation. Low diffusion yields steep gradients and localized high‑concentration microdomains.
- ER Leak vs. Channel Release – The ratio of J_leak to J_chan critically determines whether a transient calcium spike or a sustained plateau emerges. Dominant channel release produces sharp, high‑amplitude spikes that can trigger neighboring cells, whereas elevated leak leads to a more uniform elevation of cytosolic calcium.
- SERCA Pump Activity – Enhancing J_pump lowers the steady‑state cytosolic calcium level and shortens the duration of calcium transients, effectively damping wave propagation. Conversely, pump inhibition or pathological pump dysfunction results in prolonged calcium elevation, a scenario reminiscent of ER stress in neurodegenerative diseases.
The sensitivity analysis demonstrates that modest alterations in any of these parameters can dramatically reshape calcium wave morphology, underscoring the delicate balance that astrocytes maintain under physiological and pathological conditions.
The authors acknowledge several limitations. The one‑dimensional geometry neglects the complex three‑dimensional morphology of astrocytic processes and the spatial heterogeneity of organelles such as mitochondria, which can act as additional calcium buffers. Moreover, many kinetic parameters are drawn from literature averages rather than cell‑specific experimental measurements, limiting quantitative predictive power.
Future work is proposed to extend the model to three dimensions, incorporate stochastic channel gating, and couple the calcium dynamics to downstream signaling pathways (e.g., glutamate release). Integration with high‑resolution calcium imaging data would enable parameter calibration and validation, thereby transforming the current framework into a robust tool for probing astrocyte‑mediated neurophysiology and pathology.
In summary, this study successfully applies the finite volume method to a biologically grounded calcium transport model, revealing how diffusion, ER leak, channel release, and SERCA pump activity collectively dictate the spatial and temporal characteristics of astrocytic calcium waves. The results provide a valuable theoretical foundation for interpreting experimental observations and for designing interventions that modulate astrocytic calcium signaling in health and disease.
Comments & Academic Discussion
Loading comments...
Leave a Comment