Anergy in self-directed B lymphocytes from a statistical mechanics perspective
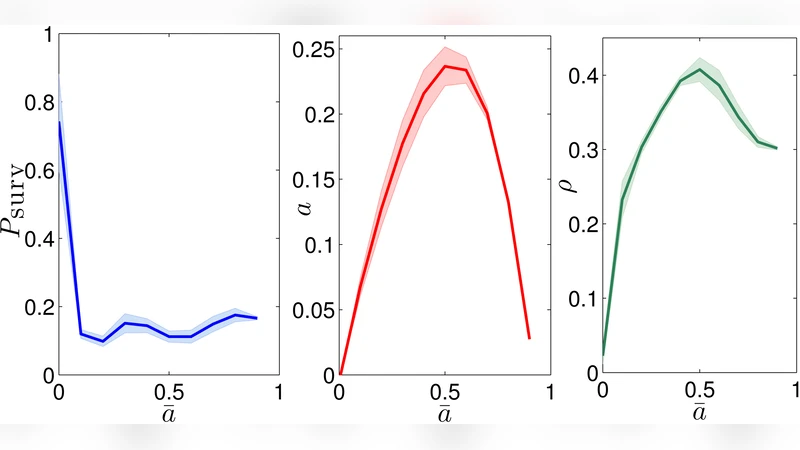
The ability of the adaptive immune system to discriminate between self and non-self mainly stems from the ontogenic clonal-deletion of lymphocytes expressing strong binding affinity with self-peptides. However, some self-directed lymphocytes may evade selection and still be harmless due to a mechanism called clonal anergy. As for B lymphocytes, two major explanations for anergy developed over three decades: according to “Varela theory”, it stems from a proper orchestration of the whole B-repertoire, in such a way that self-reactive clones, due to intensive interactions and feed-back from other clones, display more inertia to mount a response. On the other hand, according to the `two-signal model", which has prevailed nowadays, self-reacting cells are not stimulated by helper lymphocytes and the absence of such signaling yields anergy. The first result we present, achieved through disordered statistical mechanics, shows that helper cells do not prompt the activation and proliferation of a certain sub-group of B cells, which turn out to be just those broadly interacting, hence it merges the two approaches as a whole (in particular, Varela theory is then contained into the two-signal model). As a second result, we outline a minimal topological architecture for the B-world, where highly connected clones are self-directed as a natural consequence of an ontogenetic learning; this provides a mathematical framework to Varela perspective. As a consequence of these two achievements, clonal deletion and clonal anergy can be seen as two inter-playing aspects of the same phenomenon too.
💡 Research Summary
The paper tackles the long‑standing problem of how the adaptive immune system distinguishes self from non‑self, focusing on B‑cell anergy. Two dominant theories have guided the field for decades. Varela’s network theory posits that the entire B‑cell repertoire self‑organizes through extensive mutual interactions, so that self‑reactive clones become inert because they are heavily coupled to the rest of the network. In contrast, the “two‑signal model” – the prevailing paradigm – argues that B‑cells require a co‑stimulatory signal from helper T‑cells; without this second signal, self‑reactive B‑cells remain anergic.
To reconcile these views, the authors construct a disordered statistical‑mechanics model of the B‑cell–helper T‑cell system. Each B‑cell clone is represented by a binary spin σ_i, each helper T‑cell by a spin τ_μ, and the couplings J_{iμ} are drawn from a random distribution, mimicking the heterogeneous affinities that arise during ontogeny. Using replica‑symmetric mean‑field theory they derive the free‑energy functional and the self‑consistent equations for the average fields acting on B‑cells. The analysis reveals a striking dependence of the effective field h_i = Σ_μ J_{iμ} τ_μ on the B‑cell’s connectivity k_i (the number of significant interactions with other clones). Low‑connectivity clones experience a sizable positive field when helpers are active, leading to proliferation and antibody production. High‑connectivity clones, however, receive a net field that is statistically close to zero because the many random couplings cancel each other out. Consequently, these highly connected B‑cells are not driven into activation even when helpers are present; they remain in an anergic state.
This result unifies the two theories: the “inertia” described by Varela emerges naturally from the statistical cancellation of signals in a densely coupled sub‑network, while the lack of a helper signal in the two‑signal model is precisely the condition that the effective field vanishes for those clones. In other words, the same mathematical mechanism explains both the network‑based damping and the co‑stimulatory requirement.
Beyond the dynamical analysis, the authors propose a minimal topological architecture for the B‑cell repertoire. They model the repertoire as a graph where nodes are clones and edges encode cross‑reactivity or shared helper dependence. During ontogenetic learning (bone‑marrow selection), clones that bind self‑peptides with high affinity tend to acquire many connections because they share epitopes with many other clones. This process drives the emergence of a “core” of highly connected nodes that are predominantly self‑directed. The graph‑theoretic picture thus provides a concrete mathematical embodiment of Varela’s qualitative ideas.
Finally, the paper argues that clonal deletion (physical elimination of strongly self‑reactive clones) and clonal anergy (functional silencing of highly connected clones) are not competing mechanisms but complementary outcomes of the same free‑energy minimization principle. Deletion removes the most energetically costly clones, while anergy neutralizes the remainder by embedding them in a densely connected sub‑network that suppresses their response to helper signals. Together, these processes ensure robust self‑tolerance while preserving the capacity to respond to foreign antigens.
In summary, the work demonstrates that a disordered spin‑glass model can capture the essential physics of B‑cell tolerance, merges two historically distinct immunological theories, and offers a parsimonious topological framework that explains why self‑reactive clones become highly connected and consequently anergic. This unified perspective has potential implications for designing interventions in autoimmunity and for improving vaccine strategies that must navigate the delicate balance between tolerance and immunity.
Comments & Academic Discussion
Loading comments...
Leave a Comment