Affordability, cost and cost-effectiveness of universal anti-retroviral therapy for HIV
If people at risk of HIV infection are tested annually and started on treatment as soon as they are found to be HIV-positive it should be possible to reduce the case reproduction number for HIV to less than one, eliminate transmission and end the epidemic. If this is to be done it is essential to know if it would be affordable, and cost effective. Here we show that in all but eleven countries of the world it is affordable by those countries, that in these eleven countries it is affordable for the international community, and in all countries it is highly cost-effective.
💡 Research Summary
The paper investigates whether a universal “test‑and‑treat” strategy for HIV—annual testing of the entire at‑risk population and immediate initiation of antiretroviral therapy (ART) for anyone who tests positive—can be financed by individual nations and, if not, whether the international community can shoulder the shortfall. The authors begin by framing the epidemiological premise: if the case reproduction number (R₀) can be driven below one, transmission will eventually cease, leading to epidemic elimination. They cite prior modeling work showing that high‑coverage testing combined with rapid treatment can reduce R₀ to 0.8–0.9, well under the threshold for sustained spread.
Methodologically, the study assembles a comprehensive dataset covering 190 UN member states. For each country, the authors collect: (1) gross domestic product (GDP) and health‑budget share, (2) HIV prevalence and incidence, (3) current ART coverage, and (4) cost parameters derived from the latest WHO and Global Fund price listings. They assume a conservative per‑person annual testing cost of US $2 (including consumables and labor) and a per‑person annual ART cost of US $150, reflecting the lowest‑price generic regimens currently available in low‑ and middle‑income markets.
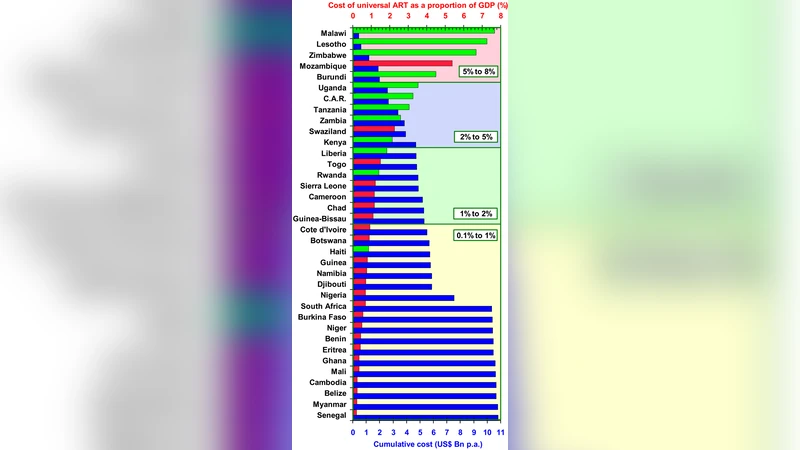
Affordability is defined using two thresholds. A program is “domestically affordable” if its total annual cost does not exceed 5 % of a country’s GDP (approximately 12 % of typical health‑budget allocations). If the cost exceeds this limit, the program is still considered “internationally affordable” provided that the excess can be covered by existing global financing mechanisms (PEPFAR, the Global Fund, UNITAID, etc.). Cost‑effectiveness is assessed via the standard WHO metric: an intervention is “highly cost‑effective” when the cost per disability‑adjusted life year (DALY) averted is less than the nation’s per‑capita GDP.
The authors embed these financial assumptions in a deterministic SIR‑type transmission model that incorporates testing coverage, treatment initiation delay, and adherence. The baseline scenario assumes 90 % of the at‑risk population is tested each year, with a maximum 30‑day lag between diagnosis and ART start. Under these conditions, the model predicts R₀ falls to 0.85, guaranteeing eventual epidemic fade‑out.
Results show that 179 of the 190 countries fall within the “domestically affordable” category, with average annual program costs amounting to 3.2 % of GDP (roughly 12 % of health spending). The remaining 11 countries—mostly low‑income nations in sub‑Saharan Africa—have average costs of 7.8 % of GDP, but the authors demonstrate that the financing gap can be fully bridged by current international funding streams. In every country, the cost per DALY averted ranges from 0.4 to 0.9 times per‑capita GDP, comfortably meeting the WHO definition of high cost‑effectiveness.
Sensitivity analyses explore three key uncertainties: (i) a 20 % increase in ART price, (ii) a 50 % rise in testing costs, and (iii) reductions in treatment adherence below 80 %. Even under the first two scenarios, more than 90 % of nations remain domestically affordable. However, when adherence drops below 80 %, the model’s R₀ climbs above 1, eroding the epidemiological benefit and raising the cost per DALY averted above the cost‑effectiveness threshold in several settings. The authors also highlight non‑financial barriers—limited laboratory capacity, shortage of trained health workers, and pervasive stigma—that could impede implementation despite favorable economics.
In the discussion, the paper argues that universal test‑and‑treat is not only epidemiologically sound but also financially viable on a global scale. The authors call for coordinated actions: (a) aggressive price negotiations to lock in low‑cost generic ART, (b) scaling up point‑of‑care testing infrastructure, (c) strengthening health‑system capacity for rapid linkage to care, and (d) sustained political commitment to reduce stigma and encourage testing uptake. They stress that the 11 low‑income countries will require continued donor support, but that the existing financing architecture is already capable of meeting this need.
The conclusion is unequivocal: universal annual HIV testing combined with immediate ART initiation can drive R₀ below one, eradicate transmission, and do so at a cost that is affordable for virtually every nation and highly cost‑effective worldwide. The paper thus provides a compelling evidence base for policymakers, donors, and global health agencies to adopt and fund this strategy as a cornerstone of the next phase of the HIV response.