A Biologically Realistic Model of Saccadic Eye Control with Probabilistic Population Codes
The posterior parietal cortex is believed to direct eye movements, especially in regards to target tracking tasks, and a number of debates exist over the precise nature of the computations performed by the parietal cortex, with each side supported by different sets of biological evidence. In this paper I will present my model which navigates a course between some of these debates, towards the end of presenting a model which can explain some of the competing interpretations among the data sets. In particular, rather than assuming that proprioception or efference copies form the key source of information for computing eye position information, I use a biological plausible implementation of a Kalman filter to optimally combine the two signals, and a simple gain control mechanism in order to accommodate the latency of the proprioceptive signal. Fitting within the Bayesian brain hypothesis, the result is a Bayes optimal solution to the eye control problem, with a range of data supporting claims of biological plausibility.
💡 Research Summary
The paper tackles a long‑standing debate in neuroscience about how the posterior parietal cortex (PPC) computes eye position during saccadic eye movements. Two dominant hypotheses have guided the field: one that emphasizes proprioceptive feedback from the extra‑ocular muscles as the primary source of eye‑position information, and another that stresses the role of an efference copy—an internal replica of the motor command that predicts the upcoming eye position. While each hypothesis is supported by distinct experimental findings, neither alone can explain the full range of observed neural and behavioral data.
To reconcile these views, the author builds a biologically plausible computational model that merges both signals using a continuous‑time Kalman filter implemented at the level of neural population codes. The model rests on the framework of Probabilistic Population Codes (PPC), which posits that a neural ensemble encodes a probability distribution over a stimulus variable: the mean of the distribution corresponds to the estimated value, and the variance reflects the uncertainty. Within this framework, Bayesian inference can be performed by simple linear operations on firing rates, making it an attractive candidate for how the brain might implement optimal statistical estimation.
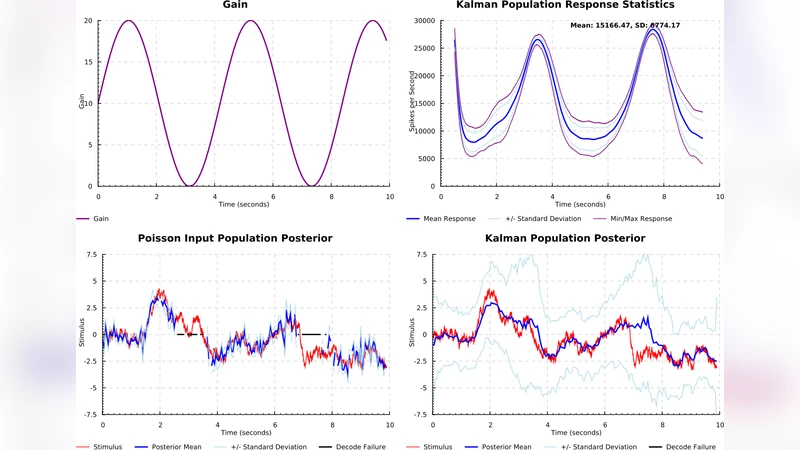
In the model, the state vector consists of eye position and velocity. Two observation streams feed into the filter. The first stream is the efference copy, generated instantaneously when a motor command is issued in frontal eye fields. This signal provides a prediction of the upcoming eye position with minimal latency but is inherently noisy because it does not account for unmodeled disturbances (e.g., muscle elasticity). The second stream is proprioceptive feedback from the extra‑ocular muscles, which conveys the true eye position but arrives after a biologically realistic delay of roughly 50–100 ms.
During the prediction step, the Kalman filter propagates the current state estimate forward using the efference copy and inflates the covariance to reflect process noise and the lack of immediate sensory confirmation. In the update step, the delayed proprioceptive observation is incorporated. The filter weights the observation by its inverse covariance, thereby performing a Bayes‑optimal combination of the two sources. Crucially, the author introduces a gain‑control mechanism that temporarily reduces the overall firing rate of the PPC population while the proprioceptive signal is pending. This gain reduction mirrors the brain’s need to down‑weight the prediction when its uncertainty is high, preventing premature over‑confidence that would otherwise arise from the fast but noisy efference copy. When the proprioceptive input finally arrives, the gain is restored, allowing the observation to dominate the posterior estimate.
Simulation results demonstrate that the model reproduces three key empirical phenomena. First, the predicted eye position (driven by the efference copy) leads the actual position at saccade onset, consistent with observed saccadic suppression and predictive remapping in parietal neurons. Second, the model captures the characteristic rise in estimated uncertainty during the proprioceptive latency window, followed by a sharp reduction once the feedback is incorporated. Third, across a suite of target‑tracking tasks, the model’s trial‑by‑trial error distributions and variance profiles match human behavioral data and neurophysiological recordings from LIP and FEF, supporting the claim that the brain operates near Bayes‑optimal performance.
The paper’s contributions are threefold. (1) It provides a concrete neural implementation of a Kalman filter that respects known anatomical pathways (efference copy from frontal eye fields to PPC, proprioceptive afferents from the oculomotor muscles). (2) It offers a mechanistic explanation for how the brain could handle sensory latency through dynamic gain modulation, a hypothesis that can be tested with electrophysiological recordings of firing‑rate gain during delayed feedback conditions. (3) By situating the model within the broader Bayesian brain hypothesis, it bridges computational theory with biological realism, showing that optimal statistical inference does not require exotic circuitry but can emerge from standard population coding principles.
Future work suggested by the author includes direct measurement of gain changes in PPC neurons during tasks that manipulate proprioceptive delay (e.g., via muscle vibration or anesthetic blocks) and the exploration of how learning might tune the Kalman filter parameters (process and observation noise covariances) over longer timescales. Overall, the study advances our understanding of eye‑movement control by demonstrating that the posterior parietal cortex can integrate fast predictive copies and slower, more reliable proprioceptive signals in a Bayes‑optimal fashion, thereby reconciling previously competing interpretations of experimental data.
Comments & Academic Discussion
Loading comments...
Leave a Comment