In Vivo Renal Clearance, Biodistribution, Toxicity of Gold nanoclusters
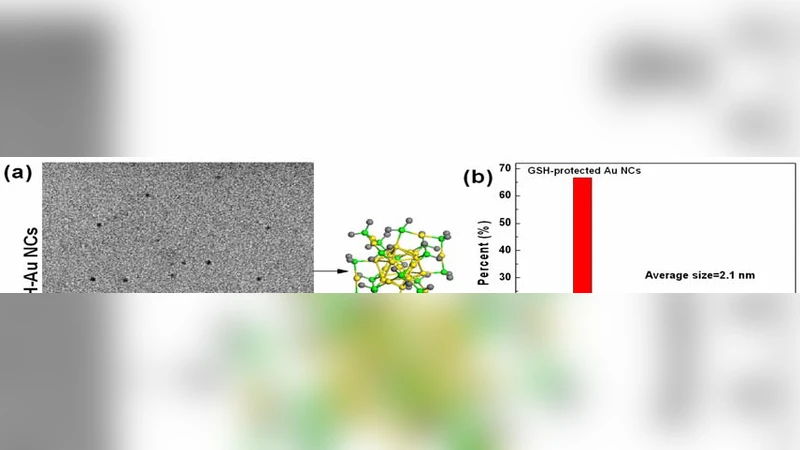
Gold nanoparticles have shown great prospective in cancer diagnosis and therapy, but they can not be metabolized and prefer to accumulate in liver and spleen due to their large size. The gold nanoclusters with small size can penetrate kidney tissue and have promise to decrease in vivo toxicity by renal clearance. In this work, we explore the in vivo renal clearance, biodistribution, and toxicity responses of the BSA- and GSH-protected gold nanoclusters for 24 hours and 28 days. The BSA-protected gold nanoclusters have low-efficient renal clearance and only 1% of gold can be cleared, but the GSH-protected gold nanoclusters have high-efficient renal clearance and 36 % of gold can be cleared after 24 hours. The biodistribution further reveals that 94% of gold can be metabolized for the GSH-protected nanoclusters, but only less than 5% of gold can be metabolized for the BSA-protected nanoclusters after 28 days. Both of the GSH- and BSA-protected gold nanoclusters cause acute infection, inflammation, and kidney function damage after 24 hours, but these toxicity responses for the GSH-protected gold nanoclusters can be eliminated after 28 days. Immune system can also be affected by the two kinds of gold nanoclusters, but the immune response for the GSH-protected gold nanoclusters can also be recovered after 28 days. These findings show that the GSH-protected gold nanoclusters have small size and can be metabolized by renal clearance and thus the toxicity can be significantly decreased. The BSA- protected gold nanoclusters, however, can form large compounds and further accumulate in liver and spleen which can cause irreparable toxicity response. Therefore, the GSH-protected gold nanoclusters have great potential for in vivo imaging and therapy, and the BSA-protected gold nanoclusters can be used as the agent of liver cancer therapy.
💡 Research Summary
This study provides a comprehensive comparative evaluation of gold nanoclusters (Au NCs) protected by bovine serum albumin (BSA) versus glutathione (GSH) with respect to renal clearance, biodistribution, and toxicity over short‑term (24 h) and long‑term (28 day) periods. The authors synthesized two distinct Au NC formulations: BSA‑protected clusters, which tend to aggregate with plasma proteins forming larger complexes, and GSH‑protected clusters, which remain ultra‑small (≈2 nm) and carry a negative surface charge that limits nonspecific protein adsorption. Using C57BL/6 mice, they administered a single intravenous dose and monitored gold content in urine, blood, major organs, and excreta by inductively coupled plasma mass spectrometry (ICP‑MS). They also measured serum biochemistry (ALT, AST, BUN, creatinine), performed histopathology, and assessed immune activation markers (CD68, CD86, cytokine panels).
Key findings include: (1) Renal clearance efficiency differs dramatically. Within 24 h, only ~1 % of the injected gold from BSA‑Au NCs is recovered in urine, whereas ~36 % of GSH‑Au NCs is cleared. By day 28, 94 % of the gold from GSH‑Au NCs has been eliminated, compared with <5 % for BSA‑Au NCs. The poor clearance of BSA‑Au NCs is attributed to the formation of larger protein‑nanocluster aggregates that exceed the glomerular filtration threshold, leading to hepatic and splenic sequestration. (2) Biodistribution mirrors clearance data. BSA‑Au NCs accumulate predominantly in liver and spleen, provoking persistent inflammation, hepatocellular injury, and histological signs of fibrosis. GSH‑Au NCs also induce an acute inflammatory response and transient kidney dysfunction (elevated BUN and creatinine) at 24 h, but these parameters return to baseline by day 28. (3) Immune profiling shows early activation of macrophages and dendritic cells for both formulations; however, only the GSH‑Au NC‑induced immune response resolves over time, whereas BSA‑Au NCs maintain elevated cytokine levels and immune cell infiltration, indicating chronic immune stimulation. (4) The authors conclude that the ultra‑small size and favorable surface chemistry of GSH‑protected Au NCs enable efficient renal excretion, dramatically reducing long‑term toxicity and making them suitable candidates for in vivo imaging, drug delivery, and theranostic applications. Conversely, BSA‑protected Au NCs, due to their propensity to form larger aggregates and persist in reticuloendothelial organs, are less appropriate for systemic use but could be repurposed as liver‑targeted agents for hepatic cancer therapy where retention is desirable.
Overall, the paper underscores the critical role of surface ligand selection and particle size in dictating nanomaterial pharmacokinetics and safety. It provides quantitative evidence that renal clearance is a viable strategy to mitigate nanomaterial toxicity and suggests that future nanomedicine design should prioritize ultra‑small, hydrophilic, and negatively charged coatings to achieve both therapeutic efficacy and biocompatibility. Further investigations in larger animal models and eventual clinical trials will be necessary to confirm translatability of these findings.
Comments & Academic Discussion
Loading comments...
Leave a Comment