Quality Control System Response to Stochastic Growth of Amyloid Fibrils
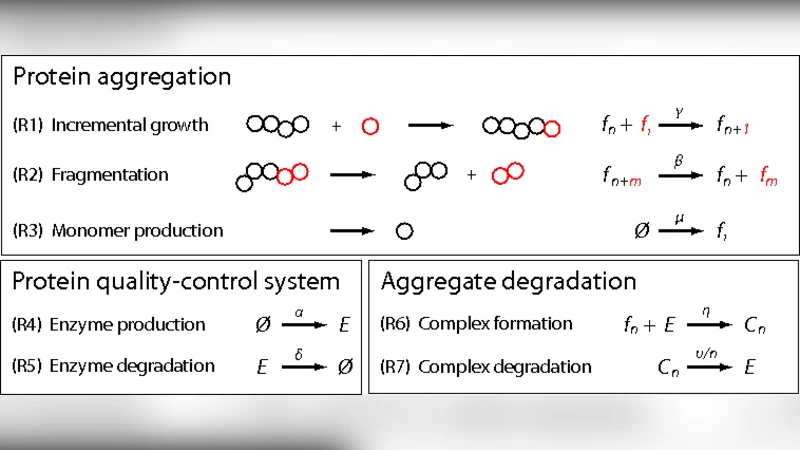
We introduce a stochastic model describing aggregation of misfolded proteins and degradation by the protein quality control system in a single cell. In analogy with existing literature, aggregates can grow, nucleate and fragment stochastically. We assume that the quality control system acts as an enzyme that can degrade aggregates at different stages of the growth process, with an efficiency that decreases with the size of the aggregate. We show how this stochastic dynamics, depending on the parameter choice, leads to two qualitatively different behaviors: a homeostatic state, where the quality control system is stable and aggregates of large sizes are not formed, and an oscillatory state, where the quality control system periodically breaks down, allowing for the formation of large aggregates. We discuss how these periodic breakdowns may constitute a mechanism for the sporadic development of neurodegenerative diseases.
💡 Research Summary
The paper presents a stochastic, single‑cell model that couples the aggregation dynamics of misfolded proteins with the activity of a protein quality‑control (QC) system. Building on established frameworks for amyloid formation, the authors model three elementary reactions for the aggregates: nucleation (formation of a seed from monomers), growth (addition of monomers to existing aggregates), and fragmentation (splitting of an aggregate into two smaller pieces). Each reaction occurs with a constant propensity (λ_nuc, λ_grow, λ_frag) and is simulated using the Gillespie algorithm, thereby capturing the inherent randomness of molecular encounters in a confined cellular volume.
The QC system is represented as an enzyme‑like entity (E) that can bind both monomers and aggregates of any size (A_n). Upon binding, the enzyme catalyzes the degradation of the bound species. Crucially, the degradation rate is assumed to decline exponentially with aggregate size: k_deg(n) = k_0 · exp(–α n). This functional form reflects experimental observations that proteasomes, lysosomes, and other clearance pathways become progressively less efficient at processing larger oligomers or fibrils. The total amount of enzyme, E_tot, is replenished at a constant synthesis rate μ, while the enzyme itself is not consumed during degradation, mimicking a catalytic turnover.
Systematic exploration of the parameter space reveals two qualitatively distinct regimes. In the “homeostatic” regime, high synthesis rate μ and/or high fragmentation propensity λ_frag keep the enzyme from saturating. Small aggregates are rapidly cleared, and the distribution of aggregate sizes settles into a low‑level steady state. The mean number of aggregates and their average size remain constant over time, indicating that the QC system successfully maintains proteostasis.
In contrast, when μ is reduced, fragmentation is slowed, or the size‑dependence parameter α is increased, the model enters an “oscillatory” regime. Initially, aggregates begin to grow; as they reach a critical size, the enzyme’s degradation capacity is overwhelmed, leading to a rapid accumulation of large fibrils. After a delay, the synthesis of new enzyme molecules restores the QC capacity, causing a swift collapse of the aggregate population. This cycle repeats, producing sustained limit‑cycle oscillations in both enzyme concentration and aggregate load. Linear stability analysis identifies a Hopf bifurcation at the boundary between the two regimes, confirming that the transition is driven by the interplay between enzyme replenishment and aggregate fragmentation.
Biologically, the oscillatory behavior offers a plausible mechanistic explanation for the sporadic onset of neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Huntington’s. Aging, oxidative stress, or genetic mutations can transiently impair the synthesis or activity of clearance pathways, pushing the cellular system into the oscillatory domain. During the “breakdown” phase, large amyloid deposits form, potentially seeding irreversible neuronal damage. Even after the QC system recovers, the damage inflicted during the high‑aggregate phase may persist, accounting for the progressive nature of these disorders despite later restoration of proteostasis.
The authors acknowledge several limitations. The model is confined to a single cell, ignoring intercellular diffusion, extracellular plaque formation, and tissue‑level feedback. Enzyme synthesis is treated as a simple first‑order process, which does not capture the complex transcriptional and post‑translational regulation of proteasome and autophagy pathways. Parameter values are chosen to illustrate qualitative behavior rather than being fitted to quantitative experimental data. Future work is suggested to embed the stochastic cell model within a multicellular lattice, incorporate realistic kinetic data for proteasomal and lysosomal degradation, and explore therapeutic interventions that modulate μ, λ_frag, or α.
In summary, this study demonstrates that stochastic aggregation coupled with size‑dependent clearance can generate either a stable proteostatic state or self‑sustained oscillations, depending on the balance of synthesis, fragmentation, and enzymatic efficiency. The oscillatory regime provides a novel theoretical framework for understanding how transient failures of the protein quality‑control system might trigger the sporadic emergence of large amyloid aggregates, thereby linking molecular stochasticity to the macroscopic pathology of neurodegeneration.
Comments & Academic Discussion
Loading comments...
Leave a Comment